Autoimmune Disease and the Thyroid: How Your Immune System Becomes Your Enemy
When the Body Attacks Itself: An Introduction to Autoimmune Disease
Few categories of illness are as fascinating — or as frustrating — as autoimmune disease. These conditions share a common, paradoxical feature: the very system designed to protect you becomes one of your greatest health threats. In autoimmune disease, the immune system mistakenly identifies the body's own tissues as foreign invaders and mounts a sustained attack against them. The consequences can range from mild inconvenience to life-altering disability, depending on which tissues are targeted and how aggressively the immune response proceeds.
Among the many organs that can fall victim to autoimmune attack, the thyroid gland holds a particularly prominent place. Autoimmune thyroid diseases collectively represent the most common of all autoimmune conditions, affecting an estimated 5% of the global population. Understanding why the immune system turns against the thyroid — and what we can do about it — is one of the most important questions in modern endocrinology and immunology.
The Biology of Autoimmunity: What Goes Wrong and Why
To understand autoimmune disease, you first need to understand how the immune system is supposed to work. The human immune system is an extraordinarily complex network of cells, proteins, and organs that work together to defend the body against infection and disease. At its core, the immune system must accomplish two seemingly contradictory tasks simultaneously: be aggressive enough to destroy dangerous pathogens, yet restrained enough to leave healthy tissue unharmed.
This balance is maintained through a process called immune tolerance. During development, immune cells are educated to recognize and ignore the body's own molecular "signatures." Cells that fail this education and would attack the self are normally eliminated through a process called clonal deletion. Additional regulatory mechanisms — including T regulatory cells (Tregs) and the production of anti-inflammatory cytokines — provide ongoing protection against self-directed immune reactions.
In autoimmune disease, one or more of these protective mechanisms fails. The exact cause varies between different autoimmune conditions and between individuals, but a combination of genetic predisposition and environmental triggers is believed to be responsible in most cases. Common environmental factors that may initiate or accelerate autoimmune disease include viral and bacterial infections, environmental toxins, dietary factors, hormonal changes, and chronic psychological stress.
The Spectrum of Autoimmune Thyroid Disease
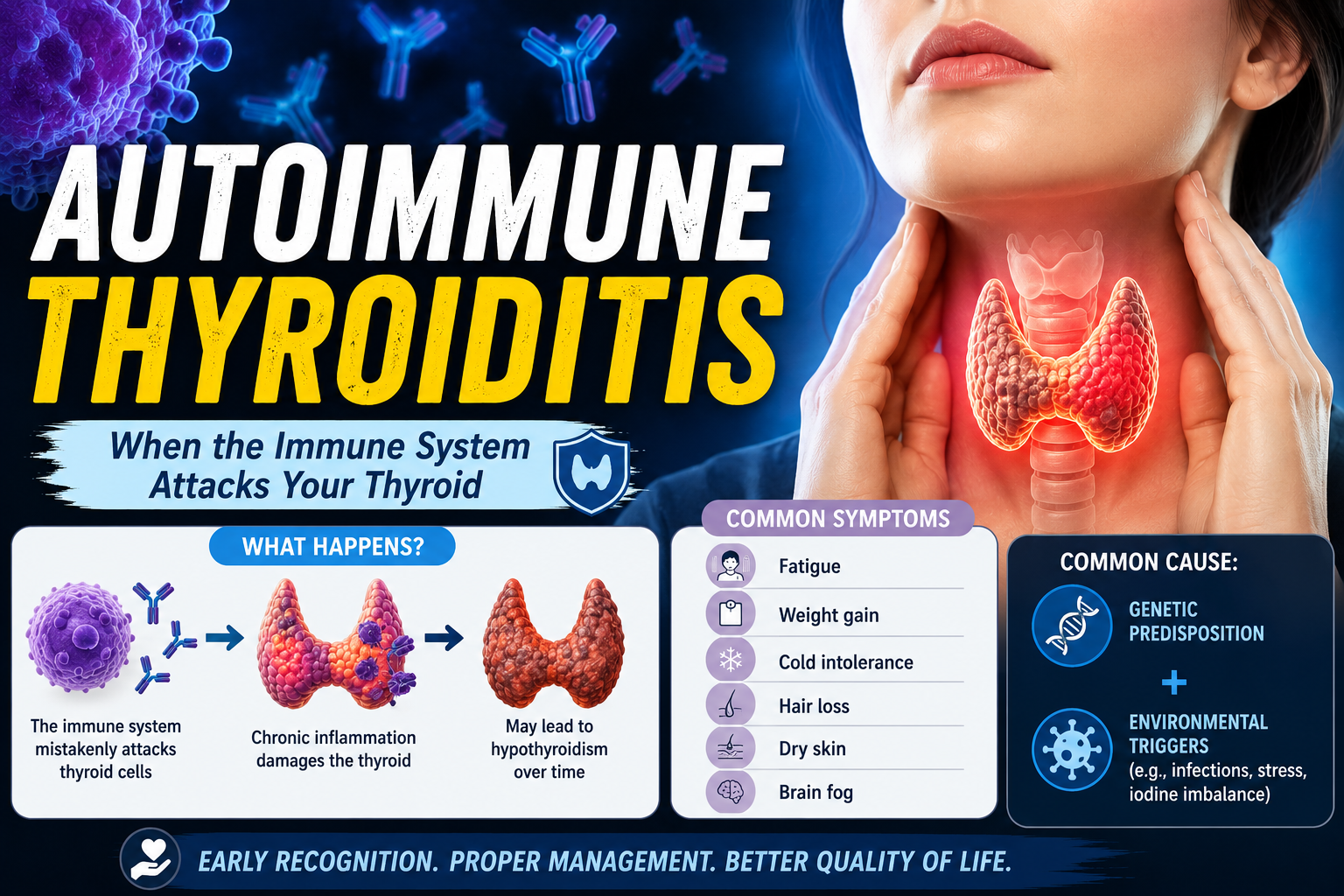
The thyroid gland is targeted in two major autoimmune conditions that sit at opposite ends of a clinical spectrum. At one end is Hashimoto's thyroiditis, in which the immune system progressively destroys thyroid tissue, ultimately leading to an underactive thyroid (hypothyroidism). At the other end is Graves' disease, in which immune antibodies over-stimulate the gland, producing an overactive thyroid (hyperthyroidism). Between these poles lie several less common autoimmune thyroid conditions, including postpartum thyroiditis, silent thyroiditis, and drug-induced thyroiditis.
What all of these conditions share is the fundamental autoimmune mechanism: an aberrant immune response directed at thyroid-specific antigens. In Hashimoto's, the primary antibodies target thyroid peroxidase (TPO) and thyroglobulin — proteins essential for thyroid hormone synthesis. In Graves' disease, the principal offending antibodies target the thyroid-stimulating hormone (TSH) receptor, causing it to be permanently activated.
The clinical course of autoimmune thyroid disease varies widely. Some individuals progress quickly through the stages of the disease, while others live with detectable antibodies for years or even decades before developing significant hormonal abnormalities. This variability underscores the importance of regular monitoring in anyone with known thyroid antibodies, even when initial thyroid function tests appear normal.
Autoimmune Disease as a Systemic Phenomenon
One of the most important — and often overlooked — aspects of autoimmune thyroid disease is that it rarely occurs in isolation. The same immune dysregulation that allows the immune system to attack the thyroid often creates vulnerability to additional autoimmune conditions. This phenomenon, known as polyautoimmunity, means that patients with one autoimmune disease have a significantly elevated risk of developing others.
Among the most common companions to autoimmune thyroid disease are celiac disease, Type 1 diabetes mellitus, rheumatoid arthritis, systemic lupus erythematosus, Sjögren's syndrome, vitiligo, pernicious anemia, and Addison's disease. This co-occurrence is not coincidental — these conditions share common genetic susceptibility loci, particularly variants in the HLA complex and in genes regulating immune checkpoints and cytokine signaling.
The implication for clinical practice is significant. A diagnosis of Hashimoto's thyroiditis or Graves' disease should prompt evaluation for common co-existing autoimmune conditions, and patients with other autoimmune diagnoses should receive regular thyroid screening. This integrated approach to care recognizes the body as an interconnected system and can lead to earlier detection and treatment of co-morbid conditions.
The Genetic Architecture of Autoimmune Thyroid Disease
Genetic factors play a substantial role in autoimmune thyroid disease susceptibility. Twin studies have demonstrated concordance rates of approximately 70% for Graves' disease and 55% for Hashimoto's thyroiditis among identical twins, indicating a strong but not absolute genetic contribution. Multiple genes have been implicated, broadly falling into two categories: immune-regulatory genes and thyroid-specific genes.
The HLA-DQ and HLA-DR alleles — located on chromosome 6 in the major histocompatibility complex (MHC) — are among the most consistently associated with both Hashimoto's and Graves' disease. Other immune genes implicated include CTLA-4 (a key regulator of T cell activation), PTPN22 (a protein tyrosine phosphatase involved in lymphocyte signaling), and CD25 (the interleukin-2 receptor alpha chain, critical for the function of regulatory T cells). Thyroid-specific susceptibility genes include the TSH receptor gene itself (particularly for Graves' disease) and thyroglobulin gene variants (particularly for Hashimoto's).
Understanding the genetic architecture of these diseases is not merely an academic exercise. It has practical implications for identifying at-risk individuals, developing targeted therapies, and ultimately potentially preventing disease onset through earlier intervention.
Hormonal Influences on Autoimmune Thyroid Disease
The striking female predominance of autoimmune thyroid disease — women are affected approximately seven to ten times more frequently than men — points strongly to the influence of sex hormones on immune function. Estrogen, in particular, appears to enhance immune reactivity and promote the production of autoantibodies. This may explain why autoimmune thyroid disease often surfaces or worsens during periods of significant hormonal fluctuation: puberty, pregnancy, the postpartum period, and menopause.
Pregnancy deserves special attention because it creates a unique immunological environment. During pregnancy, immune tolerance is deliberately broadened to prevent rejection of the genetically foreign fetus. This immune suppression often temporarily improves autoimmune thyroid conditions. However, in the months following delivery, the immune system rebounds with increased vigor — a phenomenon known as "immune rebound" or postpartum immune activation. This can trigger postpartum thyroiditis, a condition affecting up to 10% of women after childbirth, which typically involves a transient hyperthyroid phase followed by a hypothyroid phase before eventual recovery of normal function in most women.
The Gut-Thyroid-Immune Axis: An Emerging Frontier
One of the most exciting developments in the field of autoimmune disease research is the growing recognition of the gut microbiome's role in immune regulation. The trillions of microorganisms that inhabit the human digestive tract are not passive passengers — they actively train and modulate the immune system throughout life. Disruptions to the microbiome (dysbiosis) have been associated with a wide range of autoimmune conditions, including autoimmune thyroid disease.
Research has shown that patients with Hashimoto's thyroiditis tend to have altered gut microbiome compositions compared to healthy controls, with reduced diversity and a different balance of bacterial species. Whether this dysbiosis precedes or follows the development of thyroid autoimmunity remains an active area of investigation. Regardless of causation, the gut-thyroid-immune axis represents a promising therapeutic target, with interventions including dietary modification, probiotic supplementation, and treatment of intestinal permeability (sometimes called "leaky gut") being explored in clinical settings.
Environmental Triggers and Lifestyle Factors
Beyond genetics and hormones, a wide range of environmental factors have been implicated as potential triggers for autoimmune thyroid disease. Iodine is perhaps the most studied environmental variable. Both severe iodine deficiency and iodine excess appear to increase thyroid autoimmunity risk, though through different mechanisms. This has important public health implications in countries that have introduced iodine supplementation programs.
Selenium, a trace element with important antioxidant and anti-inflammatory properties, plays a critical role in thyroid hormone metabolism. Selenium deficiency has been associated with increased thyroid antibody levels and disease activity, and several randomized controlled trials have demonstrated that selenium supplementation can reduce anti-TPO antibody titers in Hashimoto's patients. Vitamin D deficiency is another commonly cited environmental factor, with numerous studies demonstrating an inverse relationship between vitamin D levels and autoimmune thyroid disease risk.
Smoking has a complex relationship with autoimmune thyroid disease. It appears to decrease the risk of Hashimoto's hypothyroidism while simultaneously increasing the risk of Graves' disease and, especially, Graves' ophthalmopathy. This paradox is thought to relate to smoking's complex effects on immune function and iodine metabolism.
Comprehensive Diagnosis: Beyond TSH Testing
The standard approach to thyroid diagnosis — measuring TSH and, if abnormal, adding free T4 and free T3 — is a good starting point, but it doesn't fully capture the autoimmune dimension of the disease. A complete autoimmune thyroid workup should include measurement of anti-TPO and anti-thyroglobulin antibodies, and in cases of suspected Graves' disease, TSH receptor antibodies (TRAb) or thyroid-stimulating immunoglobulins (TSI).
Thyroid ultrasound has become an increasingly important diagnostic tool, not only for detecting nodules but for assessing gland texture, echogenicity, and vascularity — findings that can confirm autoimmune thyroid disease even when antibodies are negative. In some patients, particularly those with Hashimoto's disease, antibodies can be seronegative, and ultrasound becomes the primary diagnostic tool.
It is worth emphasizing that having elevated thyroid antibodies without any abnormality in TSH or thyroid hormone levels — a condition sometimes called euthyroid autoimmune thyroiditis — is not a benign finding to be ignored. These individuals are at significantly elevated risk of developing overt thyroid dysfunction and deserve regular monitoring and, in some cases, preventive interventions.
Treatment Philosophy: Treating the Whole Person, Not Just the Lab Values
Perhaps the greatest challenge in managing autoimmune thyroid disease is the persistent gap between what laboratory values show and how patients actually feel. Many patients with Hashimoto's who are on thyroid hormone replacement and have TSH levels within the normal range continue to experience significant symptoms — fatigue, brain fog, weight issues, mood disturbances, and musculoskeletal pain. This disconnect has led to growing recognition that effective management of autoimmune thyroid disease requires addressing both the hormonal and the immune dimensions of the condition.
This integrative approach may include optimizing thyroid hormone levels beyond simple TSH normalization, addressing co-existing nutritional deficiencies, reducing dietary inflammatory triggers, implementing evidence-based stress reduction strategies, and in appropriate cases, using low-dose naltrexone or other immunomodulatory agents that are currently being studied for their potential benefits in autoimmune conditions.
Autoimmune disease of the thyroid is not a simple, one-dimensional condition amenable to a one-size-fits-all solution. It is a multifaceted interaction between genetics, immunity, hormones, environment, and lifestyle. Respecting this complexity is the foundation of truly effective, patient-centered care.