Autoimmune Thyroid Disease: Everything You Need to Know
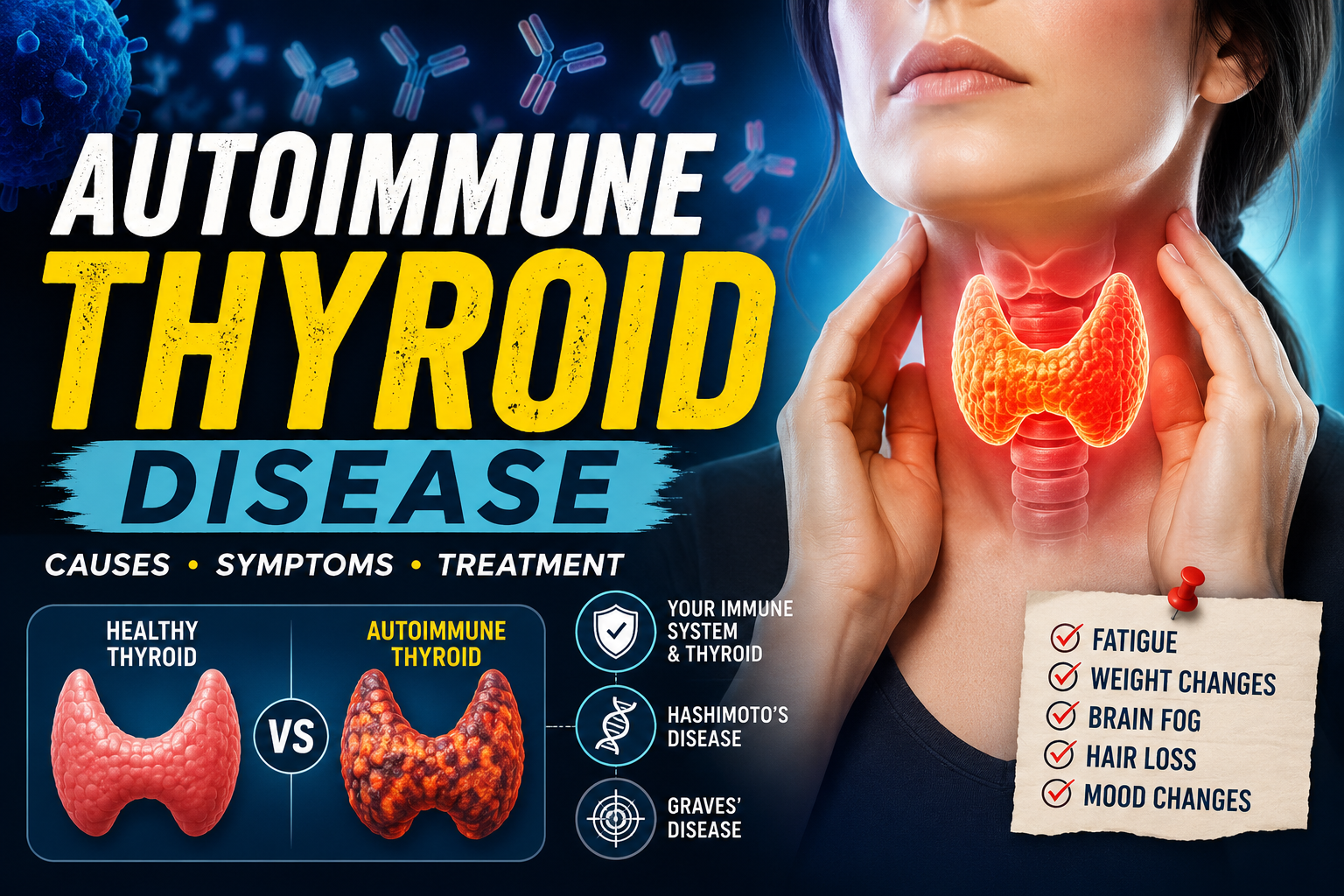
Understanding Autoimmune Thyroid Disease: What Your Body Is Trying to Tell You
Autoimmune thyroid disease is one of the most prevalent autoimmune conditions in the world, affecting millions of people — the vast majority of whom are women. Yet despite its widespread nature, it remains widely misunderstood, frequently misdiagnosed, and often undertreated. If you've recently received a diagnosis or suspect something is wrong with your thyroid, this comprehensive guide will walk you through everything you need to know about the condition, from the biological mechanisms behind it to the daily management strategies that can dramatically improve your quality of life.
What Is the Thyroid Gland and Why Does It Matter?
Before diving into autoimmune disease, it's worth understanding the organ at the center of it all. The thyroid is a small, butterfly-shaped gland located at the base of the neck, just below the Adam's apple. Despite its modest size, it plays an outsized role in virtually every system in your body. The thyroid gland produces two primary hormones: thyroxine (T4) and triiodothyronine (T3). These hormones regulate your metabolic rate, meaning they determine how quickly your cells convert nutrients into energy. But their influence doesn't stop there. Thyroid hormones affect heart rate, body temperature, digestion, muscle function, brain development, bone maintenance, and even your mood. When the thyroid is functioning normally, this biochemical symphony runs smoothly. When it doesn't, the consequences are felt throughout the entire body.
The thyroid's activity is regulated by the pituitary gland, which releases thyroid-stimulating hormone (TSH). When TSH levels are high, the thyroid is prompted to produce more T3 and T4. When levels are sufficient, TSH drops and the thyroid slows down. This feedback loop is elegant and efficient — until the immune system decides to interfere.
The Immune System: Your Body's Double-Edged Sword
In autoimmune thyroid disease, the problem doesn't originate in the thyroid itself — it originates in the immune system. Under normal circumstances, the immune system is a marvel of biological engineering. It identifies and destroys foreign invaders such as bacteria, viruses, and parasites while carefully leaving the body's own tissues alone. This self-recognition is made possible by a complex array of proteins and cells that have been trained since birth to distinguish "self" from "non-self."
In autoimmune conditions, this self-recognition system breaks down. The immune system begins to see the body's own tissues as threats and launches an attack against them. In autoimmune thyroid disease, the target is the thyroid gland. Immune cells infiltrate the gland, antibodies attack thyroid proteins, and over time, the gland becomes damaged — either becoming overactive (hyperthyroidism) or underactive (hypothyroidism).
There are two primary types of autoimmune thyroid disease: Hashimoto's thyroiditis (which leads to hypothyroidism) and Graves' disease (which leads to hyperthyroidism). While both are autoimmune in origin, their clinical presentations, antibody profiles, and treatments differ considerably.
Hashimoto's Thyroiditis: The Most Common Autoimmune Thyroid Disease
Hashimoto's thyroiditis — also known as chronic lymphocytic thyroiditis or autoimmune thyroiditis — is the leading cause of hypothyroidism in developed countries. First described by Japanese physician Hakaru Hashimoto in 1912, the disease is characterized by a progressive immune-mediated destruction of the thyroid gland. As the gland is slowly damaged, it loses its ability to produce adequate amounts of thyroid hormone, leading to the wide-ranging symptoms of hypothyroidism.
The hallmarks of Hashimoto's disease on laboratory testing are elevated levels of anti-thyroid peroxidase (anti-TPO) antibodies and/or anti-thyroglobulin (anti-Tg) antibodies. These antibodies are produced by the immune system and are directed against proteins essential for thyroid hormone production. Their presence in the blood is a diagnostic indicator that the immune system is actively attacking the thyroid.
In the early stages of Hashimoto's, the thyroid may actually go through periods of overactivity — a phenomenon sometimes called "Hashitoxicosis" — as damaged cells release stored hormone into the bloodstream. This can cause symptoms resembling hyperthyroidism before the gland eventually becomes underactive.
Graves' Disease: When the Thyroid Goes Into Overdrive
Graves' disease is the second major form of autoimmune thyroid disease and the most common cause of hyperthyroidism. Unlike Hashimoto's, where the immune system destroys the thyroid, in Graves' disease the immune system produces antibodies — called thyroid-stimulating immunoglobulins (TSI) — that mimic TSH and continuously stimulate the gland to produce excess hormone.
The result is a thyroid that is perpetually in overdrive, flooding the body with too much T3 and T4. Symptoms include rapid heart rate, significant weight loss despite normal or increased appetite, excessive sweating, heat intolerance, tremors, anxiety, and in some cases, a distinctive bulging of the eyes known as Graves' ophthalmopathy. Treatment for Graves' disease typically involves anti-thyroid medications, radioactive iodine therapy, or in some cases, surgical removal of the thyroid.
Who Gets Autoimmune Thyroid Disease? Risk Factors and Genetics
Autoimmune thyroid diseases do not affect everyone equally. Women are five to ten times more likely than men to develop autoimmune thyroid disease. This striking gender disparity suggests that sex hormones — particularly estrogen — may play a role in modulating immune responses. The risk of developing Hashimoto's disease appears to peak during periods of significant hormonal change, such as puberty, pregnancy, and menopause.
Genetics also play a significant role. If a parent or sibling has been diagnosed with Hashimoto's or Graves' disease, your risk is significantly elevated. Specific genetic variants, particularly in the HLA-DR gene region, have been associated with increased susceptibility. However, genetics alone does not determine outcome — environmental factors play a crucial role as well.
People who have one autoimmune disease are significantly more likely to develop others. Conditions such as Type 1 diabetes, rheumatoid arthritis, lupus, celiac disease, and vitiligo frequently co-occur with autoimmune thyroid disease. Chronic psychological stress, iodine intake levels, and certain infections may also trigger autoimmune thyroid disease in susceptible individuals.
Recognizing the Symptoms of Autoimmune Thyroid Disease
The symptoms of autoimmune thyroid disease vary considerably depending on whether the condition results in an underactive or overactive thyroid. In hypothyroidism (most often caused by Hashimoto's), common symptoms include: persistent fatigue and sluggishness, unexplained weight gain, cold intolerance, constipation, dry skin and brittle hair or nails, puffiness around the eyes and face, slowed heart rate, depression, poor memory and concentration (often called "brain fog"), heavy or irregular menstrual periods, and muscle weakness or aches.
In hyperthyroidism (as seen in Graves' disease), the clinical picture is nearly the opposite: rapid or irregular heartbeat, unexplained weight loss, increased appetite, heat intolerance, excessive sweating, nervousness and anxiety, difficulty sleeping, frequent bowel movements, and trembling in the hands and fingers.
Diagnosing Autoimmune Thyroid Disease
Diagnosis of autoimmune thyroid disease typically involves a combination of laboratory tests and clinical assessment. Your doctor will likely order a TSH (Thyroid-Stimulating Hormone) test as the first-line screening, followed by Free T4 and Free T3 tests to measure the levels of free thyroid hormones in the blood. Thyroid antibody tests — including anti-TPO, anti-Tg, and TSI — confirm the autoimmune origin. A thyroid ultrasound can reveal the texture, size, and vascularity of the gland.
Early and accurate diagnosis is critical because untreated thyroid disease can lead to serious complications, including heart problems, nerve damage, infertility, and in rare cases, life-threatening thyroid storm or myxedema coma.
Treatment Approaches for Autoimmune Thyroid Disease
Treatment depends on which type of autoimmune thyroid disease is present and its severity. For Hashimoto's hypothyroidism, the standard treatment is hormone replacement therapy with levothyroxine (synthetic T4). This medication is generally well-tolerated, taken once daily, and highly effective when dosed appropriately. Some patients with persistent symptoms despite normal TSH levels may benefit from the addition of liothyronine (synthetic T3).
For Graves' hyperthyroidism, treatment options include antithyroid drugs (methimazole or propylthiouracil), radioactive iodine ablation, or thyroidectomy. Beyond conventional medical treatment, many patients benefit from lifestyle modifications including an anti-inflammatory diet, stress management, optimal sleep, and management of nutrient deficiencies such as selenium, vitamin D, and zinc.
The Role of Diet and Lifestyle in Autoimmune Thyroid Disease
While medication forms the cornerstone of treatment, many patients with autoimmune thyroid disease report meaningful symptom improvements with dietary and lifestyle changes. An anti-inflammatory diet rich in vegetables, fruits, lean proteins, and healthy fats may help modulate immune activity. Some research suggests that selenium supplementation can reduce anti-TPO antibody levels in Hashimoto's patients. Gluten elimination is also widely discussed in the thyroid community, particularly given the high co-occurrence of celiac disease with Hashimoto's thyroiditis.
Stress management is equally important. Chronic stress elevates cortisol, which can suppress thyroid function and drive inflammation. Practices such as yoga, meditation, deep breathing, and regular moderate exercise have all been shown to reduce inflammatory markers and improve overall immune regulation.
Living Well With Autoimmune Thyroid Disease
A diagnosis of autoimmune thyroid disease is not a sentence to a diminished quality of life. With proper diagnosis, appropriate treatment, and thoughtful self-care, the vast majority of people with these conditions live full, active, and healthy lives. The key is working closely with a knowledgeable endocrinologist or thyroid specialist, monitoring labs regularly, and paying attention to your body's signals. Patient advocacy is equally important — learning to communicate your symptoms effectively, seeking second opinions when necessary, and staying informed about the latest research can make a profound difference in your long-term outcomes. Autoimmune thyroid disease is complex and chronic — but it is also manageable, and understanding it is the first powerful step toward reclaiming your health.

