Diverticulitis Causes: Why Does Diverticulitis Develop and What Triggers Flares?
Understanding Diverticulitis Causes: The Complete Scientific Explanation
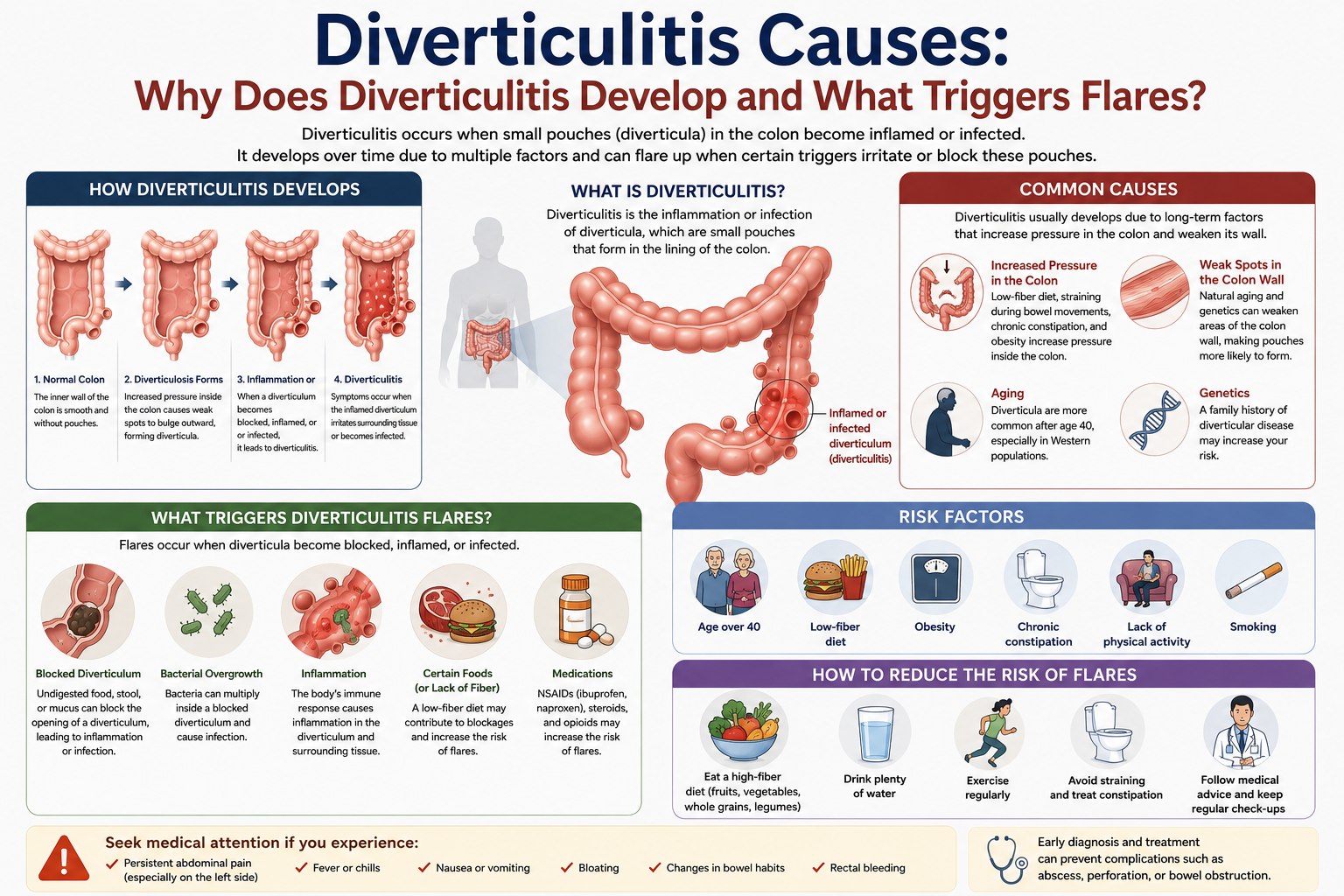
When patients receive a diagnosis of diverticulitis, one of the first questions they ask is: "Why did this happen to me?" It's a completely natural question, and understanding the underlying causes of diverticulitis is not just intellectually satisfying — it's essential for making the lifestyle changes that can prevent future episodes. Diverticulitis doesn't arise from a single cause; rather, it develops as the result of a complex interplay between anatomy, diet, gut biology, lifestyle factors, and in some cases, genetic predisposition. This comprehensive guide explores each of these contributing factors in depth, providing you with a thorough understanding of why diverticulitis develops and what you can do to reduce your risk.
The Foundation: How Diverticulitis Develops from Diverticulosis
To understand the causes of diverticulitis, you first need to understand its precursor condition: diverticulosis. Diverticulosis is the presence of small, bulging pouches called diverticula in the wall of the colon (large intestine). These pouches are extremely common, particularly in older adults in Western countries, affecting more than 50% of people over the age of 60.
Diverticulitis occurs when one or more of these diverticula become inflamed or infected. But what exactly triggers this inflammation? The traditional explanation — that small particles of food (seeds, nuts, corn) get trapped in the diverticula and cause infection — has been largely debunked by scientific research. The current understanding is considerably more nuanced and involves micro-perforations, bacterial overgrowth, altered gut microbiome, and chronic low-grade mucosal inflammation.
The two main mechanisms proposed for diverticulitis development are: (1) micro-perforation theory — a tiny hole develops in the neck of a diverticulum, allowing bacteria and intestinal contents to leak into the surrounding tissue, triggering inflammation; and (2) obstruction theory — the opening of a diverticulum becomes blocked by a small piece of hardened fecal material (fecalith), creating conditions for bacterial overgrowth and mucosal injury within the trapped pouch. Current evidence suggests that micro-perforation is the more common mechanism.

Primary Cause #1: Low Dietary Fiber Intake
The relationship between low dietary fiber intake and diverticular disease is one of the most consistent findings in gastroenterology research. Denis Burkitt, a pioneering British surgeon who worked extensively in sub-Saharan Africa in the 1970s, was among the first to formally propose the dietary fiber hypothesis. He observed that diverticular disease was virtually unknown in rural African populations consuming high-fiber, plant-based diets, while it was increasingly common in Western populations eating processed, low-fiber foods. This observation led to decades of research that has largely confirmed — with important nuances — the causal role of fiber deficiency.
How exactly does fiber deficiency cause diverticulitis? The mechanism involves multiple pathways:
Increased intracolonic pressure: When dietary fiber is inadequate, stools are small, hard, and difficult to pass. The colon must generate much higher internal pressures to propel these hardened stools through the bowel. This elevated pressure is transmitted to the colon wall over years and decades, weakening vulnerable points (particularly where blood vessels penetrate the muscular wall) and eventually causing herniation — the formation of diverticula.
Slowed intestinal transit: A low-fiber diet slows the movement of intestinal contents through the colon (prolonged transit time), increasing the time that potentially irritating materials are in contact with the colonic mucosa. This prolonged contact may increase mucosal vulnerability and the likelihood of micro-injury.
Altered gut microbiome: Dietary fiber serves as the primary food source for beneficial colonic bacteria. Without adequate fiber, the diversity and abundance of beneficial microorganisms declines, while potentially harmful bacteria may proliferate. This dysbiosis (microbiome imbalance) is increasingly recognized as a factor in diverticulitis pathogenesis.
The Nurses' Health Study and the Health Professionals Follow-Up Study — two of the largest and most methodologically rigorous epidemiological studies in medicine — both found significant inverse associations between dietary fiber intake and risk of symptomatic diverticular disease. Individuals consuming the most fiber had significantly lower risk compared to those consuming the least.
Primary Cause #2: Aging and Structural Changes in the Colon
Age is the single most powerful risk factor for diverticulosis — and consequently for diverticulitis. The prevalence of diverticulosis increases dramatically with age: from less than 10% in people under 40, to approximately 30-40% by age 60, to over 65% by age 80. This strong age dependence reflects fundamental structural changes in the colon that occur as part of normal aging.
As we age, the connective tissue matrix of the colon wall undergoes significant remodeling. Collagen — the primary structural protein of connective tissue — becomes increasingly crosslinked and less elastic with age. Studies using electron microscopy and biochemical analysis have shown that the colonic wall in older individuals is stiffer, less capable of stretching to accommodate changing intraluminal pressures, and more vulnerable to mechanical failure at weak points. This is why diverticula form preferentially at the sites where blood vessels penetrate the muscular wall — these pre-existing anatomical gaps become sites of herniation when the surrounding tissue loses its structural integrity.
Additionally, age-related changes in the enteric nervous system (the network of neurons controlling gut motility) and in smooth muscle function may contribute to abnormal motility patterns that generate excessive intraluminal pressure.
Primary Cause #3: Alterations in the Gut Microbiome
The gut microbiome — the vast community of bacteria, viruses, fungi, and archaea inhabiting the colon — is increasingly recognized as a central player in diverticulitis pathogenesis. This is an exciting and rapidly evolving area of research that may eventually lead to microbiome-based prevention and treatment strategies.
Several lines of evidence support a role for microbiome dysbiosis in diverticulitis:
Association studies: Research comparing gut microbiome profiles in patients with diverticular disease versus healthy controls has found consistent differences in microbial community composition. Patients with diverticulosis and those with recurrent diverticulitis tend to have reduced diversity, lower abundance of beneficial bacteria (such as Lactobacillus and Bifidobacterium species), and higher abundance of potentially pathogenic organisms.
Antibiotic effects: The fact that rifaximin (a poorly absorbed oral antibiotic) reduces symptoms in patients with symptomatic uncomplicated diverticular disease and may reduce the risk of diverticulitis in some populations suggests that the gut microbiome plays an active role in disease expression.
Dietary modulation: High-fiber diets promote the growth of specific bacterial taxa that produce short-chain fatty acids (SCFAs) such as butyrate, propionate, and acetate. These SCFAs are the primary fuel source for colonocytes (cells lining the colon), support mucosal barrier function, reduce inflammation, and promote healthy gut motility. Deficiency in fiber-fermenting bacteria and the SCFAs they produce may contribute to mucosal vulnerability and increased susceptibility to diverticulitis.
Post-diverticulitis microbiome changes: There is emerging evidence that episodes of acute diverticulitis themselves alter the gut microbiome in ways that may increase the risk of recurrence, potentially creating a vicious cycle.
Primary Cause #4: Physical Inactivity and Sedentary Lifestyle
Multiple large prospective studies have demonstrated a significant inverse relationship between physical activity and the risk of diverticular disease. Regular exercise appears to be protective against both the development of diverticulosis and the occurrence of diverticulitis episodes.
The mechanisms by which physical activity protects against diverticulitis are multiple:
Enhanced gut motility: Physical activity, particularly vigorous aerobic exercise, promotes intestinal peristalsis (the rhythmic muscular contractions that move contents through the bowel). Better gut motility means faster intestinal transit, reduced time for potentially harmful bacteria to accumulate, and less mechanical stress on the colonic wall.
Reduced constipation: Regular exercise is well established as a treatment for constipation. Less constipation means softer, more easily passed stools, lower intraluminal pressures, and less straining — all of which reduce pressure on the colon wall and decrease diverticula formation.
Anti-inflammatory effects: Regular moderate-intensity exercise has systemic anti-inflammatory effects, reducing circulating levels of inflammatory cytokines such as IL-6 and TNF-alpha. Given that chronic low-grade inflammation appears to play a role in diverticulitis pathogenesis, these anti-inflammatory effects may be protective.
Gut microbiome modulation: Emerging research suggests that regular physical activity positively modulates gut microbiome composition, increasing microbial diversity and the abundance of beneficial bacteria — the same bacteria whose deficiency is linked to diverticular disease risk.
The Health Professionals Follow-Up Study found that men who engaged in the highest level of vigorous physical activity had a 37% lower risk of diverticulitis compared to those who were least active. Similar findings have been reported in other large cohort studies.
Primary Cause #5: Obesity and Metabolic Dysfunction
Obesity — particularly central (abdominal) obesity — is a well-established risk factor for diverticulitis and is associated with more severe disease, more complications, and higher rates of recurrence. The mechanisms linking obesity to diverticulitis are complex and likely involve multiple pathways:
Chronic systemic inflammation: Adipose tissue, particularly visceral fat, is metabolically active and secretes a range of pro-inflammatory cytokines (adipokines) including leptin, resistin, and various interleukins. This chronic inflammatory state may lower the threshold for diverticular inflammation and impair the body's ability to resolve inflammatory episodes.
Altered gut microbiome: Obese individuals consistently show distinct gut microbiome profiles characterized by reduced diversity and altered bacterial community composition compared to lean individuals. These obesity-associated microbiome changes overlap substantially with the dysbiosis observed in diverticular disease.
Increased intraabdominal pressure: Excess abdominal fat increases intraabdominal pressure, which is transmitted to the colon wall and may contribute to diverticula formation and inflammation.
Dietary patterns: Obesity is often associated with dietary patterns characterized by high intake of processed foods, red meat, and sugar combined with low fiber intake — the same dietary pattern most strongly associated with diverticular disease risk.
A landmark study by Strate and colleagues found that both high BMI and increasing waist circumference were independently associated with significantly higher risk of diverticulitis, even after adjusting for other risk factors including physical activity and dietary fiber intake.

Primary Cause #6: Red Meat and Processed Food Consumption
Several large epidemiological studies have examined the relationship between specific dietary components (beyond fiber) and diverticulitis risk. Red meat consumption — particularly processed red meat such as hot dogs, sausages, and deli meats — has emerged as a potentially independent risk factor.
A comprehensive analysis from the Health Professionals Follow-Up Study found that men consuming the highest amounts of red meat (more than 13 servings per week) had a 58% higher risk of diverticulitis compared to those consuming the least (less than 4 servings per week). This association persisted after adjustment for dietary fiber intake, suggesting that the link between red meat and diverticulitis is not simply mediated by lower fiber content in red-meat-heavy diets.
The proposed mechanisms include: possible direct mucosal irritation from heme iron (the form of iron in red meat), effects of red meat on gut microbiome composition (high red meat intake is associated with increased abundance of bile acid-metabolizing bacteria that may produce mucosal irritants), increased production of N-nitroso compounds and heterocyclic amines (potentially mutagenic byproducts of meat cooking and digestion), and the inflammatory effects of advanced glycation end-products (AGEs) produced during high-temperature cooking of meat.
Primary Cause #7: Nonsteroidal Anti-inflammatory Drugs (NSAIDs)
Regular use of NSAIDs — including aspirin, ibuprofen, naproxen, and celecoxib — is one of the most clearly established modifiable risk factors for complicated diverticulitis. Multiple studies and systematic reviews have confirmed that regular NSAID users have a significantly higher risk of diverticular perforation, abscess formation, and other serious complications compared to non-users.
The mechanism is thought to involve NSAID-mediated inhibition of prostaglandin synthesis, which compromises the protective mucosal barrier of the intestine. Prostaglandins play a crucial role in maintaining mucosal integrity, regulating blood flow to the intestinal wall, and supporting the repair of minor mucosal injuries. When prostaglandin synthesis is chronically suppressed, the colon's defenses against bacterial invasion are weakened, increasing the likelihood that bacteria will penetrate through micro-injuries in diverticula.
Importantly, the risk associated with NSAIDs appears to be dose-dependent: higher doses and more frequent use are associated with greater risk. Even low-dose aspirin (used for cardiovascular protection) has been associated with modest increases in diverticular complication risk in some studies.
Primary Cause #8: Corticosteroids and Immunosuppressive Medications
Patients taking corticosteroids (such as prednisone) or other immunosuppressive medications for conditions like rheumatoid arthritis, inflammatory bowel disease, organ transplantation, or chronic kidney disease have significantly higher rates of complicated diverticulitis. Immunosuppression impairs the body's ability to mount an adequate local immune response against bacterial invasion, allowing what might otherwise be a contained inflammatory episode to progress to abscess formation or perforation.
Importantly, immunocompromised patients also frequently present atypically — with fewer classic symptoms such as fever and localized abdominal tenderness — making diagnosis more challenging and potentially delaying appropriate treatment. This diagnostic difficulty further contributes to worse outcomes in this population.
Primary Cause #9: Opioid and Constipating Medication Use
Opioid analgesics (such as morphine, oxycodone, hydrocodone, and codeine) significantly slow intestinal motility and cause constipation — one of the most well-known side effects of these medications. Severe opioid-induced constipation leads to hard, retained stools that dramatically increase intraluminal pressure in the colon. For patients with pre-existing diverticulosis, this sustained high intraluminal pressure creates conditions highly conducive to diverticular inflammation and micro-perforation.
Other medications that cause constipation — including certain antidepressants (particularly tricyclics), antipsychotics, calcium channel blockers, anticholinergic agents, and iron supplements — may similarly increase diverticulitis risk through constipation-mediated mechanisms.
Primary Cause #10: Genetic Predisposition
Growing evidence suggests that genetic factors contribute meaningfully to diverticular disease susceptibility. A large population-based twin study in Sweden found that approximately 40% of the variance in diverticular disease risk was attributable to genetic factors, with the remaining 60% attributable to environmental and lifestyle factors. This indicates that both genes and environment matter, and that neither alone tells the full story.
Several types of genetic variation have been proposed as contributors to diverticular disease risk:
Connective tissue gene variants: Polymorphisms in genes encoding collagen types, elastin, and matrix metalloproteinases may affect the structural integrity of the colonic wall and its susceptibility to herniation.
Immune response gene variants: Variants in genes regulating innate immune function (including toll-like receptor genes and cytokine genes) may affect the intensity of inflammatory responses to bacterial invasion of diverticula, influencing whether diverticulitis develops or remains subclinical.
Gut motility gene variants: Variants affecting the enteric nervous system and smooth muscle function may influence colonic motility patterns and intraluminal pressure generation.
It is important to note that having a genetic predisposition to diverticular disease does not mean that diverticulitis is inevitable. Genetic risk factors interact with environmental and lifestyle factors — meaning that adopting protective behaviors (high-fiber diet, regular exercise, healthy weight) may significantly reduce the expression of genetic risk.
What Triggers Individual Diverticulitis Episodes?
Understanding the underlying causes of diverticulitis (the conditions that make someone vulnerable) is distinct from understanding what triggers a specific acute episode. While the research on episode-specific triggers is less definitive, several factors have been proposed and studied:
Constipation and straining: Episodes of constipation leading to prolonged high intracolonic pressure may trigger micro-perforations in susceptible individuals. This may explain why diverticulitis episodes sometimes follow periods of dietary change, travel, or reduced physical activity.
Dietary changes: Abrupt changes in diet — particularly sudden reductions in fiber intake — may increase constipation and intracolonic pressure. Periods of dietary indiscretion (holiday eating, travel) are anecdotally associated with diverticulitis flares, though this is difficult to study prospectively.
Dehydration: Inadequate fluid intake hardens stools and worsens constipation, potentially triggering episodes in vulnerable individuals.
Stress: Psychological stress affects gut motility, gut permeability, and immune function through the gut-brain axis. While the evidence specifically linking psychological stress to diverticulitis episodes is limited, stress is a recognized trigger for many gastrointestinal conditions and may play a role here as well.
NSAID use: As discussed above, NSAID use around the time of an acute episode may precipitate or worsen diverticular inflammation through compromised mucosal defenses.
Understanding the Risk: Who Is Most Vulnerable?
Based on the evidence reviewed above, individuals most at risk for developing diverticulitis are those who combine multiple risk factors simultaneously: an older adult (over 50) who consumes a low-fiber diet, is physically inactive, has a BMI in the overweight or obese range, regularly uses NSAIDs or opioids, smokes, and has a family history of diverticular disease. This constellation of risk factors creates a "perfect storm" for diverticular disease development and progression to symptomatic diverticulitis.
Younger adults (under 50) who develop diverticulitis tend to present differently than older patients: they are more likely to be male, more likely to be obese, and are increasingly affected in a trend that parallels rising rates of obesity and poor dietary habits in younger age groups. Some research suggests that diverticulitis in younger patients may be biologically distinct, with different genetic and metabolic drivers compared to the age-related diverticulitis typical of older populations.
What You Can Do: Modifying the Modifiable Causes
While you cannot change your age or genetic makeup, many of the most important risk factors for diverticulitis are modifiable through lifestyle changes. The evidence clearly supports the following preventive strategies:
Increase dietary fiber substantially: Aim for 25-35 grams per day from diverse plant sources including fruits, vegetables, whole grains, legumes, and nuts. This is the single most evidence-based dietary intervention for diverticular disease prevention.
Exercise regularly: Aim for at least 150 minutes of moderate-intensity aerobic exercise per week, with some vigorous exercise included when possible. This reduces constipation, promotes gut motility, and has anti-inflammatory effects.
Achieve and maintain a healthy weight: Even modest weight loss in overweight or obese individuals reduces systemic inflammation and improves gut health.
Stay well hydrated: Drink at least 6-8 glasses of water daily to support fiber function and prevent constipation.
Reduce red and processed meat consumption: Replace red meat with lean poultry, fish, legumes, and plant-based proteins where possible.
Use NSAIDs judiciously: When pain relief is needed, use acetaminophen preferentially over NSAIDs. Discuss with your doctor whether any NSAID use is necessary in the context of your diverticular disease.
Quit smoking: Smoking cessation improves gut health and reduces systemic inflammation.
Conclusion
Diverticulitis is not caused by a single factor but by a convergence of multiple influences: structural vulnerabilities that develop with aging, dietary patterns that compromise gut health, lifestyle factors that increase colonic pressure and reduce mucosal defenses, microbiome alterations, genetic predispositions, and medication effects. Understanding these causes empowers patients to take targeted action. While no preventive strategy guarantees a diverticulitis-free life, adopting the evidence-based lifestyle modifications outlined in this guide can meaningfully reduce your risk and improve your overall digestive and general health. Work with your healthcare team to understand your personal risk profile and develop an individualized prevention plan.