Hashimoto's Disease: The Definitive Patient Guide to Understanding, Treating, and Thriving
What Is Hashimoto's Disease? An Evidence-Based Introduction
Hashimoto's disease is a chronic autoimmune disorder in which the body's own immune system mounts an attack against the thyroid gland, gradually impairing its ability to produce the hormones that regulate virtually every metabolic process in the human body. Named after the Japanese surgeon Hakaru Hashimoto, who first described the condition in 1912, it is now recognized as the most common autoimmune disease worldwide and the leading cause of hypothyroidism in iodine-sufficient countries.
Despite its extraordinary prevalence — affecting an estimated 1 in 20 people, with women comprising roughly 90% of diagnosed cases — Hashimoto's disease remains one of the most underdiagnosed and undertreated conditions in modern medicine. Many patients spend years navigating a confusing maze of non-specific symptoms before receiving a diagnosis. Many more receive a diagnosis but do not receive care that addresses the full complexity of the disease. This guide aims to change that — providing a thorough, evidence-based overview of Hashimoto's disease that empowers patients and informs caregivers alike.
The Anatomy of an Autoimmune Attack: How Hashimoto's Disease Develops
Hashimoto's disease is not a sudden event — it is a gradual process that unfolds over months to years before it becomes clinically apparent. The disease begins with a breakdown in immune tolerance: the immune system, which should leave the body's own tissues unharmed, begins to recognize thyroid proteins as foreign and launches an attack against them.
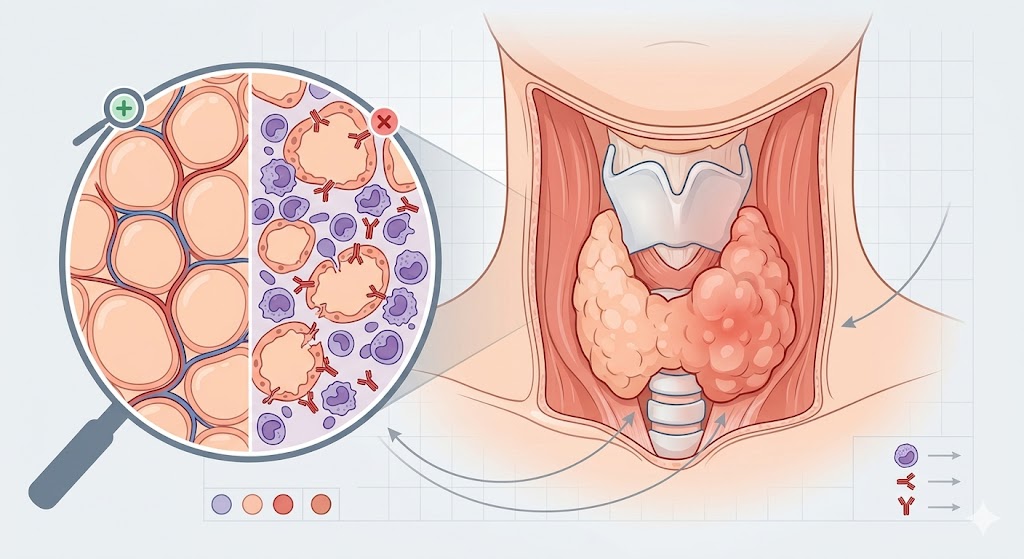
The two primary targets of the autoimmune response in Hashimoto's disease are thyroid peroxidase (TPO) — an enzyme critical for synthesizing thyroid hormones — and thyroglobulin (Tg) — the precursor protein from which T3 and T4 are assembled. Autoantibodies directed against these proteins (anti-TPO and anti-Tg) are the laboratory hallmarks of the disease, detectable in the blood of the vast majority of Hashimoto's patients. These antibodies are not merely markers — they actively amplify the destructive process by activating complement and facilitating antibody-dependent cellular cytotoxicity.
Alongside antibody-mediated damage, cellular immunity plays a central role. CD4+ T helper cells — particularly those producing the inflammatory cytokine interferon-gamma — infiltrate the thyroid gland in large numbers. This lymphocytic infiltration is so characteristic of Hashimoto's disease that it is considered a defining histological feature. Over time, the combined effect of antibody-mediated and cellular immunity-mediated damage destroys an increasing proportion of thyroid follicular cells, progressively reducing the gland's hormone-producing capacity.
Hashimoto's Disease and the Spectrum of Thyroid Dysfunction
One of the most important concepts to grasp about Hashimoto's disease is that it encompasses a broad spectrum of thyroid functional states, not a single fixed condition. The natural history of the disease typically follows a progressive course, but individual patients can occupy very different positions along this spectrum at any given time, and progression is neither linear nor inevitable.
Euthyroid Hashimoto's: In the earliest detectable phase, thyroid hormone levels (TSH, fT4, fT3) remain within normal ranges, but thyroid antibodies are elevated. This phase may last for many years. Some patients remain euthyroid indefinitely; others progress to subclinical or overt hypothyroidism. Even in the euthyroid state, many patients report symptoms — fatigue, brain fog, muscle aches — that are likely attributable to the systemic inflammatory state rather than to thyroid hormone deficiency per se.
Subclinical hypothyroidism: As the autoimmune process continues and thyroid cell mass decreases, the pituitary gland compensates by increasing TSH output — resulting in TSH above the normal range while free T4 remains normal. This is subclinical hypothyroidism. Whether and when to treat subclinical hypothyroidism remains one of the most debated questions in endocrinology. Current evidence and most guidelines recommend treatment when TSH exceeds 10 mIU/L, in patients who are pregnant or trying to conceive, and in younger symptomatic patients. For others, individual clinical judgment is required.
Overt hypothyroidism: When the gland can no longer maintain adequate hormone output despite maximal TSH stimulation, free T4 falls below normal and overt hypothyroidism is established. At this point, symptoms are typically significant and treatment with levothyroxine is unambiguously indicated. The goal of treatment is typically normalization of TSH and symptom resolution, though achieving both simultaneously requires individualized dose optimization.
Hashitoxicosis: A minority of Hashimoto's patients — particularly early in the disease course — experience transient hyperthyroidism when inflammatory destruction of thyroid tissue releases large amounts of stored thyroid hormone into the circulation. This phase, sometimes called Hashitoxicosis, can mimic Graves' disease with palpitations, weight loss, heat intolerance, and anxiety. It is self-limiting, and antithyroid drugs are generally not appropriate; beta-blockers may be used for symptom control.
The Genetics of Hashimoto's Disease: Nature's Contribution
No discussion of Hashimoto's disease is complete without acknowledging the substantial role that genetics plays in determining who develops the condition. Family studies demonstrate that first-degree relatives of Hashimoto's patients have significantly elevated risks of autoimmune thyroid disease, and twin studies show concordance rates of approximately 55% for Hashimoto's among identical twins — confirming both a strong genetic contribution and the importance of environmental factors in disease expression.
The HLA (human leukocyte antigen) complex on chromosome 6 — which encodes the proteins that present antigens to T lymphocytes — contains the most important Hashimoto's susceptibility variants. Specific HLA-DR and HLA-DQ alleles are associated with increased risk, partly because they influence how efficiently thyroid antigens are presented to the immune system. Beyond HLA, important susceptibility genes include CTLA-4 (a regulator of T cell activation), PTPN22 (involved in lymphocyte signaling), and the thyroglobulin gene itself. Genetic research using large genome-wide association studies (GWAS) continues to identify additional susceptibility loci, deepening our understanding of the molecular mechanisms underlying the disease.
Understanding the genetic architecture of Hashimoto's disease has practical implications. Family members of Hashimoto's patients — especially daughters and sisters — should be aware of their elevated risk and consider periodic thyroid antibody and function screening. They may also benefit most from proactive lifestyle measures aimed at reducing the environmental triggers that transform genetic vulnerability into manifest disease.
Co-existing Conditions: The Polyautoimmunity Pattern
Hashimoto's disease rarely travels alone. The same underlying immune dysregulation that permits the immune system to attack the thyroid creates vulnerability to additional autoimmune conditions — a phenomenon known as polyautoimmunity. Clinicians managing Hashimoto's patients should be alert to this pattern and screen for co-existing conditions that are significantly over-represented in this population.
Celiac disease is particularly important to identify, as it shares both genetic susceptibility loci and potential pathophysiological mechanisms with Hashimoto's. Studies have found celiac disease in approximately 4–6% of Hashimoto's patients, compared to approximately 1% in the general population. In patients with Hashimoto's and co-existing celiac disease, strict gluten-free diet not only treats the intestinal disease but has been shown in some studies to reduce thyroid antibody levels — suggesting a shared inflammatory driver.
Type 1 diabetes mellitus, Addison's disease, rheumatoid arthritis, Sjögren's syndrome, systemic lupus erythematosus, vitiligo, pernicious anemia, alopecia areata, and inflammatory bowel disease are all found at higher rates in Hashimoto's patients than in the general population. A comprehensive evaluation of Hashimoto's should therefore include screening for the most common co-existing conditions based on clinical presentation, family history, and laboratory findings.
Diagnosing Hashimoto's Disease: A Practical Framework
The diagnosis of Hashimoto's disease rests on three pillars: clinical assessment, laboratory testing, and imaging. No single test is definitive in isolation, and the diagnosis is most reliably established by integrating findings across all three domains.
Clinical assessment: A thorough history should document symptom onset and progression, family history of autoimmune conditions, recent significant stressors or illnesses, reproductive history, and medication use. Physical examination may reveal a goiter (thyroid enlargement) in some patients, though the gland may be normal in size or even atrophied in advanced disease. Associated physical findings (dry skin, hair loss, puffy face, slow relaxation of deep tendon reflexes) support the diagnosis of hypothyroidism.
Laboratory testing: TSH is the cornerstone of thyroid function assessment. Elevated TSH indicates hypothyroidism. Free T4 and free T3 complete the hormonal picture. Anti-TPO antibodies are elevated in approximately 90–95% of Hashimoto's cases; anti-thyroglobulin antibodies in 60–80%. Either alone is sufficient to confirm the autoimmune origin, but testing both increases sensitivity. Comprehensive assessment should also include CBC (to detect anemia), ferritin, vitamin B12, vitamin D, and consideration of screening for celiac disease.
Thyroid ultrasound: While not universally required for diagnosis when antibodies are clearly elevated, ultrasound provides valuable additional information. The characteristic sonographic appearance of Hashimoto's disease includes reduced echogenicity (the gland appears darker than normal thyroid tissue), heterogeneous echotexture, and often fibrous stranding. The gland may be enlarged in early disease or small and atrophied in long-standing disease. Importantly, ultrasound can identify nodules that require further evaluation — and in seronegative cases, ultrasound findings compatible with Hashimoto's can support the diagnosis when antibody testing is normal.
The Full Spectrum of Treatment: From Medication to Lifestyle
Effective management of Hashimoto's disease requires a multi-dimensional approach that extends well beyond prescribing levothyroxine. While thyroid hormone replacement remains the standard medical treatment for hypothyroid Hashimoto's, optimal outcomes require attention to the immune dimension of the disease, nutritional status, lifestyle factors, and — critically — the patient's subjective experience of their condition.
Levothyroxine dosing should be individualized, not standardized. The conventional target of TSH within the population reference range (typically 0.5–4.5 mIU/L) may not represent the optimal target for every individual. Many endocrinologists now advocate for a personalized approach in which the dose is adjusted to achieve both TSH normalization and symptom resolution. For patients who remain symptomatic despite optimized levothyroxine, evaluation of fT3 levels and consideration of combination T4/T3 therapy is appropriate. Desiccated thyroid extract (DTE), which contains both T4 and T3 in a natural ratio, is preferred by some patients and is a legitimate therapeutic option when patients fail to achieve adequate symptom relief on synthetic T4 alone.
Selenium supplementation at 200 mcg/day has the strongest nutritional evidence base in Hashimoto's disease management, with consistent reductions in anti-TPO antibody titers across multiple randomized trials. Vitamin D optimization, zinc sufficiency, and omega-3 fatty acid intake are additional evidence-supported nutritional considerations. Gluten elimination should be considered, particularly in patients with positive celiac disease antibodies, and may be worth a monitored trial in symptomatic seronegative patients.
Stress management deserves a central place in Hashimoto's disease management, not as a soft add-on but as a core therapeutic strategy. The HPA axis, activated by chronic psychological stress, profoundly influences immune regulation and thyroid function. Mind-body interventions — including mindfulness-based stress reduction (MBSR), yoga, progressive muscle relaxation, and cognitive-behavioral therapy — have demonstrated measurable effects on inflammatory markers and immune regulation in autoimmune conditions. Regular moderate exercise, consistent sleep schedules, and attention to work-life balance are not luxuries for Hashimoto's patients — they are therapeutic necessities.
Hashimoto's Disease in Men: The Overlooked Minority
With women comprising approximately 90% of Hashimoto's cases, men with the condition are frequently overlooked — both in clinical practice, where physicians may be less likely to consider the diagnosis, and in research, where male patients are underrepresented. Yet Hashimoto's disease in men is far from rare, and its presentation may have distinct features that warrant clinical awareness.
Men with Hashimoto's may present with subtle symptoms more attributable to testosterone deficiency — loss of libido, reduced muscle mass, erectile dysfunction — because hypothyroidism can suppress testosterone production. Cardiovascular manifestations, including elevated LDL cholesterol, hypertension, and resting bradycardia, may be more prominent in men. Fatigue and cognitive symptoms are as common in men as in women, but may be less readily attributed to a thyroid condition by clinicians who don't have the condition on their radar for male patients.
The management principles for Hashimoto's disease are broadly the same in men and women, but men deserve the same thorough evaluation and proactive management approach. Raising awareness that Hashimoto's is not exclusively a female disease is an important step toward reducing the diagnostic delay that many men with the condition experience.
Looking Forward: The Future of Hashimoto's Disease Treatment
The future of Hashimoto's disease management holds considerable promise. Several exciting research directions are moving from laboratory investigation toward clinical application. Immunotherapy approaches — including tolerogenic dendritic cell therapy, regulatory T cell expansion protocols, and antigen-specific immunotherapy — aim to address the root cause of the disease by restoring immune tolerance to thyroid antigens, rather than merely compensating for the hormonal consequences of thyroid destruction.
Microbiome-targeted therapies, including precision probiotic formulations and fecal microbiota transplantation, are being investigated for their potential to reduce autoimmune activity by restoring healthy immune regulation through the gut-immune axis. Advanced diagnostics, including thyroid antibody isotyping, cytokine profiling, and genetic susceptibility testing, are moving toward clinical availability and may ultimately enable more personalized, predictive approaches to Hashimoto's disease management.
Perhaps most fundamentally, there is a growing recognition within medicine that Hashimoto's disease — like other autoimmune conditions — requires a paradigm shift from disease management to disease modification. The goal is not merely to compensate for what the immune system has destroyed, but to interrupt the destructive process itself, preserve remaining thyroid function, and ultimately prevent the cascade from beginning at all. This is an ambitious but increasingly achievable goal, and it is driving some of the most exciting research in endocrinology and immunology today.