Hashimoto's Thyroiditis: The Science of Chronic Thyroid Inflammation and How to Heal
Hashimoto's Thyroiditis: Understanding Chronic Inflammation at the Cellular Level
Hashimoto's thyroiditis stands as one of the most compelling examples of the immune system's capacity for both protection and self-destruction. In this condition, the elaborate machinery of adaptive immunity — the T cells, B cells, antibodies, and cytokines that evolved to defend us from infection — turns its full force against the thyroid gland, creating a state of chronic, relentless inflammation that progressively compromises thyroid function over months, years, and decades.
Understanding Hashimoto's thyroiditis at the cellular and molecular level is not merely an academic exercise. It provides the conceptual foundation for everything that follows: why certain symptoms occur, why some patients progress faster than others, why conventional treatment addresses some problems but not others, and why emerging therapies targeting the immune process itself hold such promise. This article explores Hashimoto's thyroiditis with the depth and nuance the condition deserves — from molecular pathogenesis to integrative management strategies rooted in the best available evidence.
The Inflammatory Architecture of Hashimoto's Thyroiditis
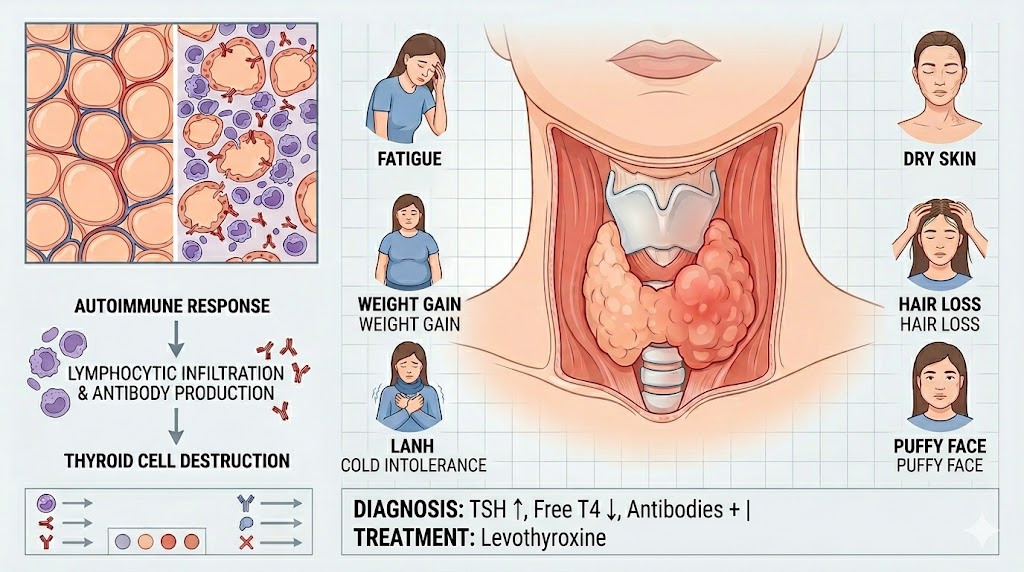
The hallmark of Hashimoto's thyroiditis on histological examination is dense lymphocytic infiltration of the thyroid gland — a cellular invasion so characteristic that it was what first struck Hakaru Hashimoto in the four patients he described in 1912. This infiltrate is not random; it has a distinct architecture that reflects the organized immune response occurring within the gland.
The infiltrate is predominantly composed of CD4+ T helper cells, CD8+ cytotoxic T cells, B lymphocytes, and macrophages. Strikingly, some of these lymphocytes organize into structures resembling lymph node germinal centers — ectopic lymphoid tissue that is formed within the thyroid itself. Within these germinal centers, B cells proliferate and mature, producing the high-affinity autoantibodies (anti-TPO and anti-Tg) that are the serological hallmarks of the disease. This local antibody production within the target organ creates a concentrated autoimmune attack that amplifies the destructive process.
The cytokine environment within the Hashimoto's thyroid is predominantly Th1-skewed — characterized by high levels of interferon-gamma (IFN-γ), tumor necrosis factor-alpha (TNF-α), and interleukin-1 (IL-1). These pro-inflammatory cytokines directly damage thyrocytes and create an environment hostile to their survival. IFN-γ, in particular, upregulates the expression of MHC class II molecules on thyrocytes — a phenomenon called "aberrant HLA-DR expression" — which may allow thyroid cells to directly present their own antigens to T cells, further amplifying the autoimmune attack. This creates a vicious cycle in which inflammation promotes antigen presentation, which promotes more inflammation.
The Natural History of Hashimoto's Thyroiditis: A Decades-Long Journey
Few chronic conditions have a natural history as variable as Hashimoto's thyroiditis. The disease can unfold over decades, with each patient following a unique trajectory determined by the interplay of genetic susceptibility, environmental exposures, hormonal fluctuations, and individual immune regulatory capacity. Understanding this natural history allows both patients and clinicians to anticipate future developments and make informed decisions about monitoring and intervention.
The earliest detectable phase of Hashimoto's thyroiditis is the presence of circulating thyroid antibodies without any abnormality in thyroid function — sometimes called the "antibody-positive euthyroid" phase. This phase can persist for years or even decades. During this time, the immune attack on the thyroid is ongoing but the gland retains sufficient reserve to maintain normal hormone output. TSH, fT4, and fT3 remain within normal limits. Many people in this phase are entirely asymptomatic; others report fatigue, brain fog, or other non-specific symptoms that may be attributable to the systemic inflammatory burden rather than thyroid hormone deficiency.
As the immune attack continues and thyroid cell mass progressively decreases, compensatory mechanisms eventually become inadequate. The pituitary gland responds to declining thyroid output by increasing TSH secretion — maintaining fT4 within the normal range at the cost of elevated TSH. This is the subclinical hypothyroidism phase. The rate of progression from this stage to overt hypothyroidism is approximately 2–5% per year in antibody-positive individuals, but is highly variable. Some patients remain subclinically hypothyroid for decades without ever developing overt hypothyroidism; others progress within a few years.
Overt hypothyroidism — defined by elevated TSH and low fT4 — represents the stage at which most patients first receive a diagnosis and begin treatment. At this point, the thyroid's reserve capacity has been exhausted and hormone production is genuinely insufficient to meet the body's needs. Without treatment, overt hypothyroidism progressively worsens and can ultimately result in myxedema — a severe, life-threatening form of hypothyroidism characterized by mental obtundation, hypothermia, bradycardia, and respiratory depression.
The Extrathyroidal Manifestations of Hashimoto's Thyroiditis
While the thyroid is the primary target of the autoimmune process in Hashimoto's thyroiditis, the condition has meaningful effects that extend well beyond the gland itself. These extrathyroidal manifestations reflect both the systemic consequences of thyroid hormone deficiency and the direct effects of immune dysregulation on other organs and systems.
The cardiovascular system is significantly affected by Hashimoto's hypothyroidism. Thyroid hormone deficiency impairs myocardial contractility and relaxation, slows heart rate, and increases peripheral vascular resistance — collectively raising the workload on the heart and increasing cardiovascular risk. Dyslipidemia, particularly elevated LDL cholesterol and triglycerides, is a well-established consequence of hypothyroidism and contributes to accelerated atherosclerosis. Diastolic dysfunction — impaired relaxation of the heart muscle between beats — is detectable even in subclinical hypothyroidism and may contribute to exercise intolerance and shortness of breath. These cardiovascular effects are largely reversible with adequate thyroid hormone treatment.
The musculoskeletal system is frequently affected in Hashimoto's thyroiditis. Muscle weakness, myalgia (muscle pain), and elevated creatine kinase (a marker of muscle damage) are common. In severe hypothyroidism, myopathy can cause significant functional impairment. Joint pain, stiffness, and carpal tunnel syndrome are also more common in Hashimoto's patients than in the general population. The exact mechanisms are multifactorial, involving both the metabolic effects of thyroid hormone deficiency and the direct inflammatory effects of the autoimmune process on musculoskeletal tissues.
The kidneys and fluid balance are affected through several mechanisms. Hypothyroidism impairs kidney filtration (reduced GFR), promotes sodium and water retention, and increases the risk of hyponatremia. The puffiness and edema that many Hashimoto's patients experience reflects not just water retention but also the accumulation of hyaluronic acid and other glycosaminoglycans in connective tissues — a process called myxedema that gives the skin its characteristic doughy texture.
The nervous system is profoundly dependent on thyroid hormone at every stage of life. In adults with Hashimoto's, the neurological manifestations range from the ubiquitous cognitive symptoms (brain fog, memory impairment, slowed processing) to the rare but serious Hashimoto's encephalopathy — a steroid-responsive encephalopathy characterized by seizures, altered consciousness, tremors, and psychosis. The pathophysiology of Hashimoto's encephalopathy remains incompletely understood but is believed to involve immune-mediated damage to cerebral vasculature and neurons.
Advanced Diagnostics in Hashimoto's Thyroiditis
Standard thyroid function testing and antibody measurement remain the foundation of Hashimoto's thyroiditis diagnosis, but advances in diagnostic technology are expanding the toolkit available to clinicians and patients. Understanding these advanced options helps patients ask the right questions and make more informed decisions about their own evaluation.
High-resolution thyroid ultrasound with elastography is increasingly used in Hashimoto's management. Standard ultrasound assesses gland size, echogenicity, and texture. Elastography adds information about gland stiffness — a measure of fibrosis — which correlates with disease severity and may predict future progression to hypothyroidism. Color Doppler ultrasound assesses gland vascularity; in the inflammatory phases of Hashimoto's, increased blood flow may be detected, reflecting heightened immune activity.
Fine-needle aspiration biopsy (FNAB) is not routinely required for the diagnosis of Hashimoto's thyroiditis but is essential when thyroid nodules are detected on ultrasound. Hashimoto's patients have a somewhat elevated rate of thyroid nodule formation, and an increased risk (though still absolute low) of papillary thyroid cancer and primary thyroid lymphoma. Any nodule meeting size or ultrasound criteria for evaluation should undergo FNAB, regardless of the Hashimoto's diagnosis.
Emerging laboratory tests include anti-TPO antibody isotyping (which subtypes may have different clinical significance), thyroid-stimulating hormone receptor antibody testing (to exclude concurrent Graves' disease in patients with mixed presentations), and cytokine profiling. Genetic testing for DIO2 polymorphisms — which affect the conversion of T4 to T3 — is gaining clinical relevance in guiding decisions about combination T4/T3 therapy, as patients with certain DIO2 variants may have suboptimal T3 levels despite adequate T4 replacement.
Integrative Approaches to Hashimoto's Thyroiditis: Beyond the Prescription Pad
Conventional medicine offers effective hormonal management of Hashimoto's thyroiditis, but many patients feel that this approach addresses the downstream consequences of the disease without touching its root cause — the dysregulated immune response. This recognition has driven growing interest in integrative and functional medicine approaches that aim to address the immune dimension of the disease directly.
The autoimmune protocol (AIP) diet represents one of the most structured dietary interventions proposed for autoimmune conditions including Hashimoto's thyroiditis. It is a phased elimination protocol that removes grains, legumes, dairy, eggs, nightshades, nuts, seeds, and refined foods in the initial phase, then systematically reintroduces foods to identify personal triggers. While large randomized controlled trials are lacking, observational studies and patient-reported outcomes suggest that a significant proportion of Hashimoto's patients experience reduced antibody levels and improved symptoms on AIP. The protocol is challenging to implement and maintain, and professional nutritional guidance is recommended.
Mind-body medicine is increasingly recognized as a legitimate component of autoimmune disease management. Mindfulness-based stress reduction (MBSR), a structured 8-week program developed by Jon Kabat-Zinn, has demonstrated measurable effects on inflammatory markers (including reduced IL-6 and CRP) in multiple clinical trials. Yoga combines physical activity with breath regulation and meditative focus in ways that appear to reduce stress hormone levels and modulate immune function. Regular practice of these techniques over months may produce meaningful reductions in disease activity in addition to improving psychological resilience and quality of life.
Acupuncture and traditional Chinese medicine approaches to thyroid conditions are increasingly studied in clinical research. Small trials suggest that acupuncture may reduce thyroid antibody levels and improve quality of life in Hashimoto's patients, possibly through effects on autonomic nervous system regulation and hypothalamic-pituitary function. While the evidence base remains limited, these approaches offer a low-risk complementary option for patients who find benefit from them.
The Role of the Micronutrient Environment in Hashimoto's Thyroiditis
The thyroid gland is one of the most micronutrient-sensitive organs in the body, and deficiencies in several key nutrients can both impair thyroid function and accelerate autoimmune disease activity. Comprehensive assessment and optimization of micronutrient status is therefore a central component of integrative Hashimoto's thyroiditis management.
Selenium is the most evidence-supported nutrient in Hashimoto's thyroiditis. The thyroid gland contains the highest concentration of selenium per gram of tissue of any organ in the body — a reflection of the critical role of selenoproteins in thyroid hormone synthesis (thioredoxin reductases and deiodinases) and in protecting thyroid cells from oxidative damage. Multiple randomized controlled trials have demonstrated that selenium supplementation at 200 mcg/day significantly reduces anti-TPO antibody titers, reduces markers of thyroid inflammation, and improves subjective well-being in Hashimoto's patients. The mechanism is believed to involve enhanced activity of glutathione peroxidases and thioredoxin reductases, which neutralize the hydrogen peroxide generated during thyroid hormone synthesis and reduce the oxidative stress that drives inflammation.
Iodine is both essential and potentially problematic in Hashimoto's thyroiditis — a paradox that requires careful clinical navigation. Iodine is an essential component of thyroid hormones, and severe iodine deficiency impairs thyroid function. However, excess iodine has been shown to trigger or exacerbate autoimmune thyroiditis in genetically susceptible individuals, possibly by increasing the immunogenicity of thyroglobulin. For patients with Hashimoto's thyroiditis who are taking supplemental iodine at high doses (e.g., in kelp supplements or high-dose iodine protocols), reduction to physiological levels is often advisable.
Iron is frequently deficient in women with Hashimoto's thyroiditis, particularly those with heavy menstrual periods. Iron deficiency impairs thyroid peroxidase activity and reduces the efficiency of thyroid hormone synthesis. Importantly, iron deficiency can worsen hypothyroid symptoms and can impair the efficacy of levothyroxine treatment. All women with Hashimoto's should have ferritin levels checked, and iron supplementation should be prescribed when deficiency is identified — while avoiding taking iron supplements within 4 hours of levothyroxine, as iron impairs its absorption.
Living With Hashimoto's Thyroiditis: Building Long-Term Resilience
Managing Hashimoto's thyroiditis over the long term requires not just the right medical regimen but the right mindset, habits, and support systems. The disease is chronic and, in the current state of medicine, not curable — but it is eminently manageable, and many people with Hashimoto's thyroiditis live full, vibrant, and productive lives. The key is approaching the condition not as a defeat but as an invitation to build a healthier relationship with your body, your healthcare, and your daily life choices.
Regular monitoring is the cornerstone of long-term management. TSH and free T4 should be checked at least annually once the condition is stable on treatment, and more frequently during periods of change — pregnancy, significant illness, medication changes, or significant weight fluctuation. Learning to recognize the symptoms of under- and over-treatment — and communicating changes promptly to your healthcare provider — is an invaluable skill. Keeping a symptom journal can help identify patterns and changes that might otherwise be difficult to articulate at a medical appointment.
The Hashimoto's community is a remarkable resource. Patient advocacy organizations, online communities, published patient memoirs, and podcasts by and for Hashimoto's patients provide a wealth of practical wisdom, emotional support, and scientific information. Being well-informed, actively engaged in your own care, and connected to others who understand your experience are not merely nice-to-haves — they are factors that research consistently shows to be associated with better health outcomes in chronic illness. Hashimoto's thyroiditis is a lifelong journey, but it is one that millions of people are walking with courage, knowledge, and growing vitality every day.