Inflamed Pancreas: Everything You Need to Know About Pancreatic Inflammation
What Does It Mean to Have an Inflamed Pancreas?
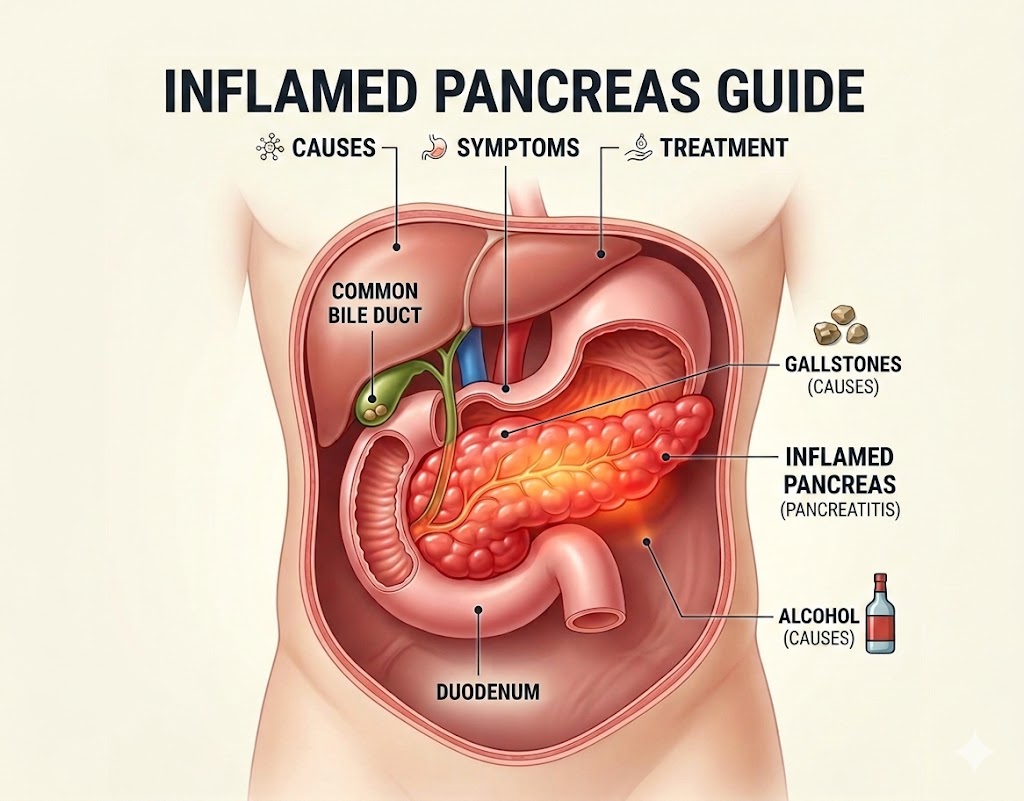
The phrase "inflamed pancreas" describes a condition in which the pancreas — a vital glandular organ located in the upper abdomen behind the stomach — undergoes an abnormal inflammatory response. This inflammation is medically referred to as pancreatitis, and it can occur in two distinct clinical forms: acute pancreatitis (sudden inflammation that resolves with treatment) and chronic pancreatitis (long-standing inflammation that causes progressive, irreversible damage). Understanding what happens at the cellular and physiological level when the pancreas becomes inflamed helps explain why this condition can be so severe and why prompt medical care is essential.
The pancreas is uniquely vulnerable to self-injury. It produces powerful digestive enzymes — including trypsin, chymotrypsin, lipase, amylase, and elastase — that are capable of digesting proteins, fats, and carbohydrates. Under normal conditions, these enzymes are stored in inactive precursor forms (called zymogens) and are only activated after they reach the small intestine. When this protective mechanism breaks down — due to any of a number of triggers — the enzymes become activated within the pancreatic tissue itself. The result is autodigestion: the pancreas begins to digest itself, triggering an inflammatory response that can range from mild swelling to catastrophic tissue destruction (necrosis).
This guide takes a comprehensive look at the inflamed pancreas — from its underlying biology and triggers to its symptoms, diagnostic evaluation, medical and surgical treatment options, complications, and long-term management. Whether you are a patient seeking to understand your own condition, a caregiver supporting someone with pancreatitis, or simply a health-conscious reader, this guide provides the knowledge needed to understand one of the most complex and important gastrointestinal conditions.
The Biology of Pancreatic Inflammation
When the pancreas becomes inflamed, a complex biological cascade is set in motion. The central event is the premature activation of trypsinogen — the inactive precursor of trypsin, one of the most powerful proteolytic (protein-digesting) enzymes in the body — within the pancreatic acinar cells (the enzyme-producing cells of the pancreas). Activated trypsin then activates other zymogens in a chain reaction, including chymotrypsin, elastase, phospholipase A2, and lipase. The activated phospholipase A2 is particularly destructive, as it breaks down the cell membranes of pancreatic cells, releasing arachidonic acid metabolites that drive further inflammation.
The inflammatory cascade that follows involves the release of pro-inflammatory cytokines — signaling molecules including interleukin-1 (IL-1), IL-6, IL-8, and tumor necrosis factor-alpha (TNF-α) — from damaged pancreatic cells and infiltrating immune cells. These cytokines amplify local inflammation and, in severe cases, spill into the systemic circulation, causing what is known as a systemic inflammatory response syndrome (SIRS). SIRS can lead to distant organ dysfunction — particularly affecting the lungs, kidneys, and cardiovascular system — even when the primary injury remains localized to the pancreas.
Simultaneously, the inflammation triggers increased vascular permeability, causing fluid to leak from the blood vessels into the spaces around the pancreas and throughout the abdominal cavity. This fluid shift — which can involve several liters of fluid in severe cases — leads to edema of the pancreas and surrounding tissues, hypovolemia (reduced circulating blood volume), and a drop in blood pressure. The combination of systemic inflammation and hypovolemia is the primary mechanism behind the hemodynamic instability that characterizes severe acute pancreatitis.
How an Inflamed Pancreas Looks on Imaging
Modern medical imaging has transformed our ability to visualize the inflamed pancreas and assess the severity of damage. On a computed tomography (CT) scan — the gold standard imaging modality for pancreatitis — an inflamed pancreas typically appears enlarged and edematous (swollen), with indistinct borders due to surrounding inflammation and fluid. The peripancreatic fat (fatty tissue surrounding the pancreas) shows signs of inflammatory infiltration, appearing hazy or stranded. Fluid collections may be visible around or within the pancreas.
In more severe cases, contrast-enhanced CT (CECT) reveals areas of pancreatic necrosis — regions of the pancreatic tissue that have lost their blood supply and are dying. These areas appear as zones of non-enhancement (dark areas) within the normally bright, contrast-enhancing pancreatic tissue. The extent of pancreatic necrosis is quantified using the CT Severity Index (CTSI), also known as the Balthazar Score, which combines assessment of overall inflammation grade and the percentage of the pancreas that is necrotic. Higher CTSI scores correlate with greater morbidity, longer hospital stays, and higher mortality rates.
Magnetic resonance imaging (MRI) and magnetic resonance cholangiopancreatography (MRCP) offer additional information, particularly regarding the integrity of the pancreatic duct and the presence of stones or strictures in the bile duct — information that is critical for planning endoscopic or surgical intervention. Endoscopic ultrasound (EUS) provides high-resolution images of the pancreas and surrounding structures and is particularly useful for detecting small gallstones (microlithiasis) and early changes of chronic pancreatitis not visible on cross-sectional imaging.

Symptoms of a Swollen and Inflamed Pancreas
The symptoms produced by an inflamed pancreas are primarily a consequence of its anatomical position, its role in digestion, and the inflammatory cascade triggered by pancreatic injury. The dominant symptom is severe upper abdominal pain — typically located in the epigastric region and often described as a deep, constant, boring or gnawing ache that may radiate to the back. The pain is generally severe enough to prompt emergency care, is exacerbated by eating (which stimulates pancreatic enzyme secretion), and may be partially relieved by leaning forward or assuming the fetal position.
Nausea and vomiting are nearly universal accompaniments to the pain of an inflamed pancreas. Unlike the vomiting associated with gastroenteritis or food poisoning, vomiting in pancreatitis does not relieve the pain — the pain source is the inflamed pancreas, not the stomach or intestine. Abdominal distension and tenderness on examination are also typical findings, reflecting both local inflammatory edema and the associated ileus (bowel paralysis) caused by the nearby inflammatory process.
Fever is present in many cases, reflecting the systemic inflammatory response. A low-grade fever (100–102°F) is common in mild pancreatitis, while higher fever with rigors may indicate a secondary infection such as infected pancreatic necrosis. Tachycardia (elevated heart rate) results from pain, fever, and fluid shifts. In severe cases, patients may develop signs of hemodynamic instability — falling blood pressure, pallor, sweating, and rapid shallow breathing — signaling the need for immediate intensive care management.
Jaundice (yellowing of the skin and eyes) occurs when inflammation or a gallstone obstructs the common bile duct, which passes through the head of the pancreas. This is most common in gallstone-related pancreatitis and indicates biliary obstruction requiring specific interventions. Two rare but important physical signs — Cullen's sign (bruising around the navel) and Grey Turner's sign (bruising along the flanks) — indicate hemorrhagic pancreatitis and carry a serious prognosis.
Diagnosing an Inflamed Pancreas
The diagnosis of a clinically inflamed pancreas is established through a combination of clinical history, physical examination, laboratory tests, and imaging. The revised Atlanta Classification criteria require at least two of three features: characteristic abdominal pain; elevated serum lipase or amylase (at least three times the upper limit of normal); and characteristic imaging findings on CT, MRI, or ultrasound. In most cases, the first two criteria are sufficient for diagnosis, and imaging is reserved for cases of diagnostic uncertainty or to assess severity and complications.
Laboratory testing is the cornerstone of biochemical diagnosis. Serum lipase is the preferred marker due to its higher specificity and longer elevation compared to amylase. Additional laboratory tests provide information about etiology and severity: serum bilirubin and liver enzymes (ALT, AST, alkaline phosphatase) are elevated in gallstone-related pancreatitis; a serum triglyceride level above 1,000 mg/dL indicates hypertriglyceridemia as the cause; an elevated white blood cell count, C-reactive protein (CRP), and procalcitonin reflect the degree of systemic inflammation; elevated creatinine and BUN indicate renal impairment; and arterial blood gas analysis may reveal respiratory compromise.
Several validated scoring systems are used to predict the severity of an inflamed pancreas and identify patients at high risk for complications. The Ranson Criteria assess 11 variables across admission and the first 48 hours. The BISAP Score uses five variables (BUN, impaired mental status, SIRS, age, and pleural effusion) scored at admission. The APACHE II and APACHE III systems provide more granular severity assessment but require more data points. Persistent organ failure for more than 48 hours is the key feature defining severe acute pancreatitis in the revised Atlanta Classification.

Treatment of an Inflamed Pancreas
Treatment of the inflamed pancreas is primarily supportive, focused on managing symptoms, preventing and treating complications, addressing the underlying cause, and optimizing the conditions for healing. The management approach differs between mild, moderately severe, and severe disease.
Fluid resuscitation is the most critical and time-sensitive intervention in acute pancreatitis. The inflammatory process causes massive fluid shifts from the intravascular space into the tissues and peritoneal cavity, leading to hypovolemia that can cause organ failure if not corrected promptly. Current guidelines recommend aggressive intravenous fluid therapy within the first 24–48 hours, using lactated Ringer's solution, which has been shown in clinical trials to reduce inflammation compared to normal saline. The goal is to achieve adequate hydration as reflected by urine output of at least 0.5 mL/kg/hour and normalization of hemodynamic parameters.
Pain management is a central component of supportive care. Opioid analgesics — including morphine, hydromorphone, and fentanyl — are the most effective pain medications for the severe pain of acute pancreatitis and should not be withheld due to concerns about worsening the sphincter of Oddi spasm, a historical concern that is not well-supported by current evidence. Non-opioid adjuncts including intravenous acetaminophen and non-steroidal anti-inflammatory drugs (NSAIDs) may be used in patients without contraindications. Epidural analgesia is an option in specialized centers for patients with severe, refractory pain.
Nutritional support has evolved significantly in recent years. The traditional approach of "pancreatic rest" — keeping patients nil by mouth (NPO) for prolonged periods to reduce pancreatic stimulation — has been largely abandoned based on evidence showing that early enteral nutrition (feeding through a nasogastric or nasojejunal tube) reduces infectious complications, organ failure, and length of hospital stay compared to total parenteral nutrition (intravenous feeding). Current guidelines recommend starting enteral feeding within 24–48 hours of admission in patients who cannot tolerate oral intake.
Managing Complications of Pancreatic Inflammation
The inflamed pancreas can give rise to a range of local and systemic complications, each requiring specific management. Acute peripancreatic fluid collections are common early findings that typically resolve spontaneously without intervention. Pseudocysts — walled-off fluid collections that persist beyond four weeks — may require drainage if they cause symptoms such as pain, gastric outlet obstruction, or infection. Drainage is typically performed endoscopically (through the stomach wall under EUS guidance), radiologically (via a percutaneous catheter), or surgically.
Walled-off pancreatic necrosis (WOPN) is a serious complication defined as a collection of necrotic pancreatic tissue encased by a well-defined inflammatory wall, developing more than four weeks after onset of necrotizing pancreatitis. Sterile WOPN can often be managed conservatively, but infected WOPN — presenting with sepsis, fever, and clinical deterioration — requires intervention. The modern approach favors a step-up approach, beginning with minimally invasive endoscopic or percutaneous drainage and escalating to more invasive surgical necrosectomy only if less invasive measures fail.
Pancreatic fistula — an abnormal connection between the pancreatic duct and another structure (such as the peritoneal cavity, pleural space, or skin) — can develop as a consequence of disruption of the main pancreatic duct. This complication may cause persistent ascites, pleural effusion, or drainage from an external fistula and typically requires endoscopic or surgical intervention to seal the ductal disruption.

Chronic Inflamed Pancreas: Long-Term Consequences
When the pancreas is repeatedly or continuously inflamed over months and years, it undergoes irreversible structural changes that define chronic pancreatitis. The repeated episodes of inflammation and autodigestion lead to progressive fibrosis — replacement of functional pancreatic tissue with scar tissue — resulting in the permanent loss of both exocrine (digestive enzyme-producing) and endocrine (hormone-producing) functions. This has profound consequences for the affected individual's health, nutrition, and quality of life.
Exocrine pancreatic insufficiency (EPI) occurs when the loss of enzyme-producing tissue reduces enzyme output below the level needed for adequate digestion. Without sufficient lipase and other enzymes, dietary fat is not absorbed properly, leading to steatorrhea (fatty, foul-smelling stools), abdominal bloating, flatulence, and progressive malnutrition. EPI is managed with pancreatic enzyme replacement therapy (PERT) — oral capsules containing concentrated digestive enzymes taken with every meal and snack. With appropriate PERT dosing, most patients achieve substantial improvement in digestion and nutritional status.
Endocrine pancreatic insufficiency — specifically, diabetes mellitus caused by destruction of the insulin-producing beta cells in the pancreatic islets — develops in up to 80% of patients with chronic pancreatitis over time. This form of diabetes, sometimes called pancreatogenic or type 3c diabetes, has distinct features from type 1 and type 2 diabetes: it is associated with simultaneous deficiency of glucagon (produced by alpha cells), which makes affected patients particularly vulnerable to severe hypoglycemia (low blood sugar). Management requires careful glucose monitoring and individualized insulin regimens.
Lifestyle Modifications for a Healthy Pancreas
For patients who have experienced pancreatic inflammation, and for those who wish to reduce their risk of developing it, several evidence-based lifestyle modifications can significantly protect pancreatic health. Eliminating or substantially reducing alcohol consumption is the most important intervention for alcohol-related pancreatitis; continued drinking after an episode of pancreatitis dramatically increases the risk of recurrence and progression to chronic pancreatitis.
Dietary changes support pancreatic recovery and long-term health. A low-fat diet reduces the stimulation of pancreatic enzyme secretion and minimizes the workload on an already stressed gland. Small, frequent meals — five to six small meals per day rather than three large ones — are better tolerated than large, fatty meals. Adequate hydration is important, particularly during recovery from an acute episode. In patients with hypertriglyceridemia-induced pancreatitis, a very-low-fat diet combined with avoidance of refined carbohydrates, alcohol, and fructose-rich beverages is essential for triglyceride control.
Smoking cessation is strongly recommended for all patients with pancreatitis. Cigarette smoking has been identified as an independent risk factor for both acute and chronic pancreatitis and significantly accelerates the progression of chronic pancreatitis to its end-stage. The mechanisms involve direct toxic effects of cigarette smoke components on pancreatic tissue, promotion of oxidative stress, and impairment of pancreatic blood flow. Quitting smoking at any stage of the disease can slow progression and reduce the risk of pancreatic cancer — a feared long-term complication of chronic pancreatitis.
Conclusion
An inflamed pancreas represents one of the most complex and potentially severe conditions in gastroenterology, encompassing a spectrum from mild, self-limiting acute episodes to progressive chronic disease with profound systemic consequences. Understanding the biology of pancreatic inflammation, recognizing the symptoms that demand urgent attention, and knowing the range of treatment options available empowers patients to seek timely care and make informed decisions about their health. With advances in early fluid resuscitation, minimally invasive management of complications, enzyme replacement therapy, and personalized nutritional support, the outcomes for patients with pancreatitis have improved substantially. Addressing modifiable risk factors — above all, alcohol use, gallstones, and hypertriglyceridemia — remains the most powerful tool available for preventing this serious condition.