Pancreas Pain: A Complete Guide to Understanding, Locating, and Treating It
Introduction: What Is Pancreas Pain and Why Does It Matter?
Pain originating from the pancreas is among the most intense, debilitating, and clinically significant forms of abdominal pain that a person can experience. Unlike many other types of abdominal discomfort that fluctuate in intensity or resolve quickly on their own, pancreas pain is typically severe, constant, and associated with a range of other alarming symptoms that signal the urgent need for medical evaluation. Yet despite its intensity, pancreas pain is frequently misidentified, delayed in diagnosis, or confused with other common abdominal conditions — a problem that can have serious consequences.
The pancreas is a deeply positioned organ, lying behind the stomach in the retroperitoneal space of the upper abdomen. Because of this hidden location, the pain it generates has a distinctive character that differs from the pain of more anteriorly positioned organs like the stomach or intestine. Understanding the unique features of pancreas pain — where it is felt, how it radiates, what makes it better or worse, and what it means clinically — is essential knowledge for patients, caregivers, and anyone who wants to be informed about their own health.
This comprehensive guide explores every dimension of pancreas pain: its anatomical basis, the spectrum of conditions that cause it, the diagnostic approach to identifying its source, the medical and interventional treatments available to manage it, and practical strategies for living with pancreatic disease. Whether you are experiencing pancreas pain for the first time or managing a long-standing pancreatic condition, the information in this guide will help you understand your body and make empowered decisions about your care.
The Anatomy Behind Pancreas Pain
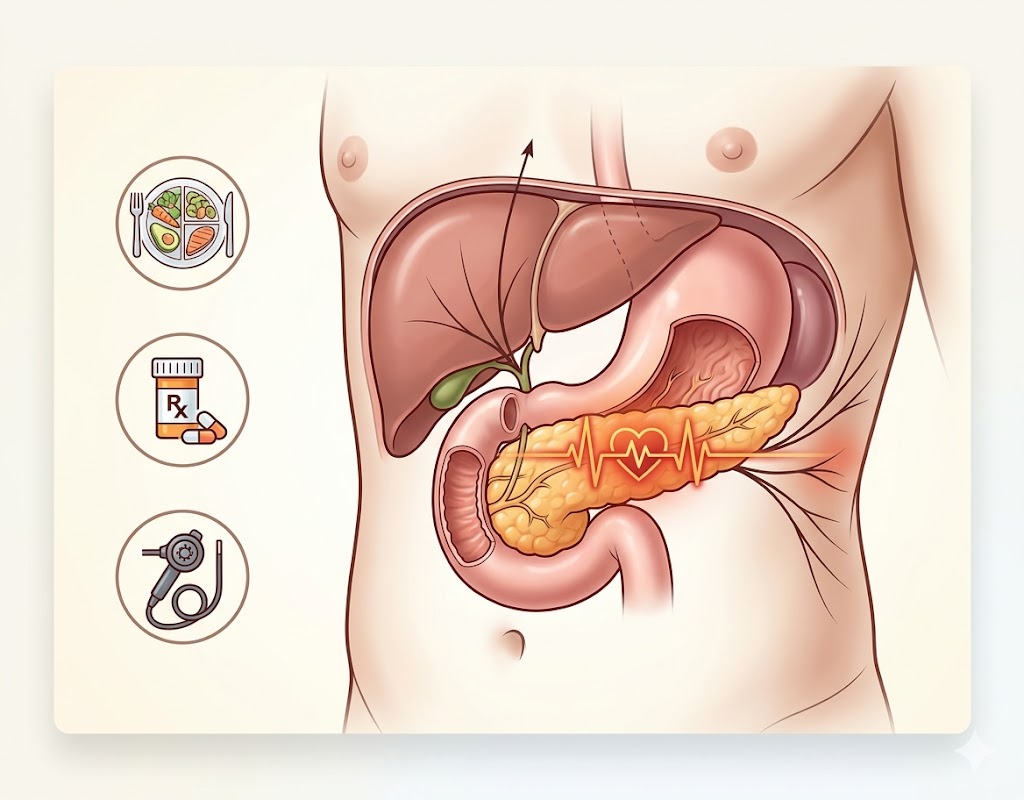
To understand why pancreas pain feels the way it does, it is helpful to understand where the pancreas sits within the body and how it relates to the surrounding anatomy. The pancreas is a soft, elongated glandular organ approximately 15–20 cm (6–8 inches) in length. It is divided into four anatomical regions: the head (the widest part, nestled in the curve of the duodenum — the first segment of the small intestine), the neck, the body (the central portion running across the upper abdomen), and the tail (the narrowest part, extending toward the spleen on the left side of the abdomen).
The entire organ lies in the retroperitoneum — behind the peritoneal cavity that contains most of the digestive organs — and lies directly in front of the vertebral column. This position is what gives pancreas pain its characteristic quality: because the pancreas is so deeply positioned and tightly associated with the posterior abdominal wall and major nerves, inflammation or disease of the pancreas tends to generate pain that is felt deep within the abdomen and radiates to the back, rather than being perceived as a surface-level or easily localized pain.
The pancreas is innervated by the celiac plexus — a large network of nerve fibers in the upper abdomen that transmits pain signals from multiple abdominal organs including the stomach, liver, small intestine, and pancreas. The convergence of pain signals from multiple organs at the celiac plexus level helps explain why pancreas pain can be difficult to localize precisely and why it often feels "deeper" and more diffuse than pain from organs closer to the abdominal surface. In cases of severe or chronic pancreatic disease, interventions targeting the celiac plexus (celiac plexus block or neurolysis) can be highly effective for pain management.

Where Is Pancreas Pain Located?
Pancreas pain is most consistently felt in the epigastric region — the central upper abdomen, below the breastbone (sternum) and above the navel. This location corresponds roughly to the position of the pancreatic body, which is the portion of the organ most centrally located in the upper abdomen. In many patients, the pain feels as though it is coming from deep within the body rather than from the surface of the abdomen, reflecting the retroperitoneal position of the pancreas.
The left upper quadrant (below the left ribcage) is frequently involved, particularly when inflammation affects the pancreatic tail or when the surrounding tissues and the nearby spleen become involved in the inflammatory process. Right upper quadrant involvement is more common when the pancreatic head — through which the bile duct passes — is affected, or when gallstones are the underlying cause. Some patients experience the pain as diffuse across the entire upper abdomen, making it difficult to localize to a single quadrant.
The radiation of pancreas pain to the back is one of its most characteristic and diagnostically important features. In approximately 50% of cases, the pain extends from the upper abdomen to the mid-back, particularly between the shoulder blades or to the area of the spine corresponding to the thoracic-lumbar junction (around the T12–L2 vertebral level). This occurs because the pancreas is in close anatomical proximity to the retroperitoneal nerves and muscles of the posterior abdominal wall. The band-like quality of this pain — wrapping from front to back — is a classic description that immediately suggests pancreatic disease to experienced clinicians.
The Character of Pancreatic Pain: What It Feels Like
Beyond its location, the qualitative character of pancreas pain is highly specific and can be distinguished from other types of abdominal pain based on several key features. Patients consistently describe pancreas pain as severe — typically rating it 7–10 on a 10-point pain scale when it is at its worst. The onset can be sudden and dramatic (particularly in acute pancreatitis triggered by gallstones or alcohol) or more gradual (in chronic pancreatitis where pain accumulates over hours).
The pain is almost universally described as constant and unrelenting rather than intermittent or cramping. This distinguishes pancreas pain from the colic of bowel obstruction or kidney stones, which typically comes in intense waves separated by periods of relative relief. Pancreatic pain, once established, persists with minimal fluctuation until treated or until the episode begins to resolve naturally.
Common descriptors used by patients to characterize pancreas pain include: boring, gnawing, pressing, aching, and crushing. Many patients say the pain feels as though something heavy is pressing down on the upper abdomen or as though a band is squeezing tightly around the midsection. The pain is deeply visceral in quality — perceived as coming from inside the body rather than from the skin, muscle, or abdominal wall.
Position significantly affects the experience of pancreas pain. Lying flat on the back (supine position) typically intensifies the pain, because this position increases pressure on the inflamed retroperitoneal pancreas against the vertebral column. Many patients instinctively find relief by sitting forward, leaning over, or assuming the fetal position (curled on the side with the knees drawn up to the chest). This positional relationship is clinically recognized as a useful indicator of retroperitoneal organ involvement and is specifically sought during clinical evaluation.
Eating and Pancreas Pain: The Food Trigger
One of the most important and practically significant features of pancreas pain is its close relationship to food intake. Eating stimulates the production and secretion of pancreatic digestive enzymes, which in the setting of an inflamed or diseased pancreas can intensify the pain dramatically. This is why patients with both acute and chronic pancreatitis universally experience worsening pain during or after meals.
In acute pancreatitis, the relationship is straightforward: eating is avoided because it acutely worsens pain, and the standard of care has historically involved temporary fasting (though current evidence now supports early enteral nutrition). In chronic pancreatitis, the relationship is more nuanced but equally impactful: patients develop postprandial pain (pain after eating) that can be so severe and predictable that they begin to avoid eating altogether. This food avoidance, combined with the malabsorption caused by pancreatic enzyme deficiency, leads to severe unintentional weight loss, malnutrition, and vitamin deficiencies — a cycle that profoundly worsens quality of life and long-term prognosis.
High-fat meals are particularly potent triggers for pancreatic pain, as fat stimulates the release of cholecystokinin (CCK) — a hormone that powerfully stimulates pancreatic enzyme secretion. Patients with chronic pancreatitis or a history of acute pancreatitis are typically advised to follow a low-fat diet as a cornerstone of pain management, aiming for no more than 20–30 grams of total fat per day during periods of disease activity.

Distinguishing Pancreas Pain from Other Abdominal Pain
Because the upper abdomen is home to many organs — including the stomach, liver, gallbladder, duodenum, spleen, kidneys, and pancreas — the differential diagnosis of upper abdominal pain is broad. Correctly identifying pancreas pain and distinguishing it from other conditions is essential for directing appropriate treatment.
Gallbladder and biliary pain (biliary colic or cholecystitis) is one of the most important conditions to distinguish from pancreatitis, as it frequently coexists with it (gallstones can cause both). Biliary colic typically produces right upper quadrant pain that radiates to the right shoulder or the right side of the back, often comes on suddenly after a fatty meal, lasts 30 minutes to several hours, and then resolves. Unlike pancreas pain, biliary colic may partially resolve between episodes. Cholecystitis (gallbladder inflammation) causes more persistent right upper quadrant pain with local tenderness (Murphy's sign) and fever. Measurement of serum lipase distinguishes pancreatitis from uncomplicated biliary disease.
Peptic ulcer disease — particularly a perforated or penetrating peptic ulcer — can mimic pancreas pain closely. Both conditions cause severe upper abdominal pain, and a penetrating posterior ulcer may radiate to the back in a similar pattern to pancreatic pain. The presence of a history of ulcer disease, use of NSAIDs or aspirin, relief of pain with antacids, and the results of upper endoscopy help differentiate. A CT scan can identify free air in the abdomen in the case of perforation.
Aortic dissection or aortic aneurysm rupture is a life-threatening emergency that can present with severe tearing back pain and should always be in the differential diagnosis of sudden severe upper abdominal or back pain, particularly in elderly patients with hypertension or known aneurysm. The pain of aortic dissection is typically described as tearing, ripping, or knife-like, often most intense at its onset, and may be accompanied by asymmetric blood pressures in the arms. CT angiography is diagnostic.
Medical Management of Pancreas Pain
Managing pancreas pain requires a comprehensive approach tailored to whether the pain is acute (in the context of an episode of acute pancreatitis) or chronic (in the setting of chronic pancreatitis). The goals of pain management differ between these two scenarios but share certain fundamental principles: adequate analgesia, nutritional optimization, and addressing the underlying cause.
In acute pancreatitis, pain management is part of the broader supportive care approach. Intravenous opioid analgesics — including morphine, hydromorphone, and fentanyl — are the most effective options and should be administered promptly via patient-controlled analgesia (PCA) or scheduled dosing for severe pain. The previous concern that morphine worsens pancreatitis by causing spasm of the sphincter of Oddi is not supported by contemporary clinical evidence, and morphine is used safely and effectively in this setting. Intravenous acetaminophen and NSAIDs (such as ketorolac) are useful adjuncts that can reduce opioid requirements. Adequate intravenous fluid resuscitation also reduces pain by improving pancreatic perfusion.
In chronic pancreatitis, pain management is significantly more complex and challenging. A stepped approach is recommended, beginning with the least aggressive interventions and escalating when simpler measures are insufficient. The first step involves dietary modification (low-fat diet, small frequent meals, avoidance of alcohol) and non-opioid analgesics (acetaminophen, NSAIDs). Pancreatic enzyme replacement therapy (PERT) can reduce pain in some patients with exocrine insufficiency by suppressing cholecystokinin release through a negative feedback mechanism. Adjuvant medications including tricyclic antidepressants, gabapentin, and pregabalin can modulate pain perception and are valuable components of a multimodal pain regimen.
Interventional Pain Management for Chronic Pancreatic Pain
When conservative medical measures fail to provide adequate relief from pancreas pain, interventional procedures offer important alternatives. These range from endoscopic techniques to neurolytic procedures and surgical options, and they are most effective when selected based on the specific anatomy of the individual patient's disease.
Endoscopic therapy — performed via ERCP (endoscopic retrograde cholangiopancreatography) — can relieve pancreatic duct obstruction caused by strictures (narrowings) or stones, which are common in chronic pancreatitis and can cause or exacerbate pain by increasing ductal pressure. Endoscopic sphincterotomy, ductal stone extraction, balloon dilation of strictures, and placement of plastic or metal stents to maintain duct patency are all tools in the endoscopic armamentarium. In carefully selected patients, these procedures can provide substantial and durable pain relief.
Celiac plexus block and celiac plexus neurolysis involve the injection of analgesic (block) or ablative (neurolysis) agents into or around the celiac plexus — the nerve network that transmits pain signals from the pancreas. Performed under CT or EUS guidance, these procedures can provide excellent pain relief in approximately 50–70% of patients. The effect of celiac plexus block is temporary (weeks to months), while neurolysis with alcohol or other agents can produce more durable relief. The procedure is generally well-tolerated, though serious complications — including orthostatic hypotension, diarrhea, and rare neurological complications — can occur.
Surgical options for chronic pancreatic pain include decompressive procedures (designed to relieve ductal hypertension in patients with dilated pancreatic ducts) such as the Puestow procedure (lateral pancreaticojejunostomy), and resective procedures (designed to remove diseased tissue) such as the Whipple procedure (pancreaticoduodenectomy) for disease predominantly in the pancreatic head, or distal pancreatectomy for tail-dominant disease. Total pancreatectomy with islet autotransplantation (TPIAT) — in which the pancreas is removed and the patient's own insulin-producing islet cells are transplanted into the liver — has emerged as an option for patients with intractable pain from hereditary or idiopathic pancreatitis who have not responded to other interventions.

Pancreas Pain and Mental Health
The impact of chronic pancreas pain on mental health is profound and deserves dedicated attention in any comprehensive discussion of this condition. Chronic pain of any kind is strongly associated with depression, anxiety, and reduced quality of life, and pancreatic pain — with its severity, unpredictability, and association with food (one of life's most fundamental pleasures) — is particularly disruptive. Studies in patients with chronic pancreatitis consistently report high rates of clinical depression (estimated at 30–50%), anxiety disorders, social isolation, occupational disability, and reduced quality of life compared to both healthy controls and patients with other chronic gastrointestinal conditions.
The relationship between pain and psychological distress is bidirectional: not only does chronic pain cause depression and anxiety, but psychological distress amplifies the experience of pain through central sensitization mechanisms, reducing pain thresholds and increasing pain-related disability. This means that addressing the psychological dimensions of pancreatic pain is not optional or secondary — it is an integral part of effective pain management. Multidisciplinary pain programs that integrate medical, nutritional, psychological, and rehabilitation services produce better outcomes than any single-modality approach alone.
Cognitive-behavioral therapy (CBT) has strong evidence for reducing pain-related disability and improving psychological wellbeing in chronic pain conditions, including chronic pancreatitis. Mindfulness-based interventions, acceptance and commitment therapy (ACT), and group pain management programs are additional evidence-based options. Ensuring that patients with chronic pancreatic pain have access to mental health support — ideally integrated into their gastroenterological care team — is an important quality standard that many healthcare systems are still working to meet.
Living with Pancreas Pain: Practical Guidance
For patients managing chronic pancreatic disease and recurring pancreas pain, practical day-to-day strategies can make a significant difference in quality of life. Dietary management is central: adopting a genuinely low-fat diet (not merely "healthier" eating, but a specific therapeutic diet limiting fat to 20–30 grams per day or less during flares) consistently reduces pain frequency and severity in many patients. Eating smaller, more frequent meals rather than large ones reduces the magnitude of pancreatic stimulation per meal and minimizes postprandial pain.
Strict avoidance of alcohol is non-negotiable for patients with alcohol-related pancreatitis and is strongly recommended for all patients with any form of pancreatitis. Even moderate alcohol consumption has been shown to increase the risk of acute attacks, accelerate the progression of chronic pancreatitis, and worsen pain in patients with established disease. If alcohol use disorder is a contributing factor, formal addiction treatment and ongoing support (including medications such as naltrexone or acamprosate) should be part of the care plan.
Smoking cessation is equally critical. Smoking is an independent risk factor for chronic pancreatitis, accelerates its progression, independently increases the risk of pancreatic cancer, and worsens the pain of established disease. Nicotine replacement therapy, varenicline (Chantix), and bupropion are effective pharmacological aids for cessation. Staying well hydrated, particularly during and after physical activity, supports pancreatic health. Gentle aerobic exercise — walking, swimming, cycling — can be beneficial when pain is controlled, as physical activity has anti-inflammatory effects and improves mood and overall wellbeing.
When to Seek Medical Help for Pancreas Pain
While chronic pancreatic pain requires ongoing management, certain presentations of pancreas pain should prompt immediate emergency medical evaluation. Seek emergency care if you experience: sudden, severe upper abdominal pain that is the worst pain you have ever felt; upper abdominal pain accompanied by fever, chills, or jaundice; pancreatic pain associated with uncontrolled vomiting that prevents fluid intake; signs of shock (severe dizziness, fainting, rapid pulse, very low blood pressure); difficulty breathing or chest pain; or confusion, altered consciousness, or extreme weakness.
These features may indicate severe acute pancreatitis, infected pancreatic necrosis, or other life-threatening complications that require urgent hospitalization and aggressive management. Even for patients familiar with their own chronic pancreatitis and accustomed to managing pain at home, a significant change in the character, location, or severity of pain — or the development of new systemic symptoms — warrants prompt medical evaluation rather than home management.
Conclusion
Pancreas pain is a complex, multifaceted medical problem that touches virtually every aspect of a patient's life — physical, nutritional, psychological, and social. Understanding its anatomical basis, characteristic features, triggering factors, diagnostic evaluation, and the full spectrum of management options — from dietary modification and medications to endoscopic interventions and surgery — is essential for both patients and healthcare providers. Advances in imaging, endoscopy, minimally invasive surgery, and pain medicine have substantially improved our ability to diagnose and treat pancreatic pain, though much work remains to be done, particularly in ensuring equitable access to multidisciplinary care. If you are experiencing pancreas pain, do not accept inadequate management as inevitable — effective treatments exist, and with the right team and the right approach, significant improvement in pain control and quality of life is achievable.